Every young chemist attending first semester organic chemistry lectures learns about benzene, the archetypical aromatic compound, in which the cloud of six π electrons is smoothly distributed along the six-membered ring and all carbon–carbon bonds are equivalent and intermediate in length between single and double bonds.
Early Discoveries
The discovery of benzene in 1825 and studies of its reactivity pushed chemists of the day to develop new theories of bonding and the concept of aromaticity was discovered. Practical-minded chemists noted the intense colors of related aromatic molecules, a consequence of the electronic structure and the difference in the energies of occupied and unoccupied molecular orbitals. They developed dyes and pigments that were among the first products of the nascent chemical industry.
Aromatic chemistry has developed steadily over the years and the current cutting edge is far removed from these low-tech beginnings. Aromatic compounds are no longer defined as being like benzene, in which the π system is a closed conjugated loop. Compounds and materials in this category may have an extended π-electron system—linear, planar, or in three dimensions. As Nazario Martín, a leader in this area, put it, “The only restrictions on these compounds is the imagination of the chemist.”
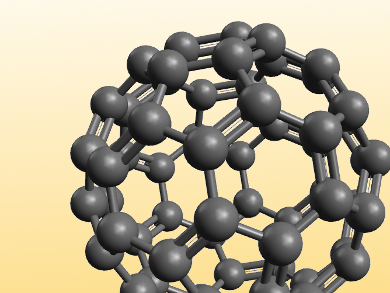
The field received a tremendous boost after the discovery of buckminsterfullerene, C60, in 1985. Related carbon structures, as well as graphene can be considered aromatic systems. And not surprisingly, chemists have also jumped on the bucky bandwagon. Nazario Martín and his group at Universidad Complutense de Madrid, Spain, are pursuing the use of fullerenes and other carbon-based materials for transforming sunlight into electricity by means of organic photovoltaic cells, and more recently, in perovskite solar cells. Curiously, his group has also reported a tridecafullerene formed by 13 C60 units and decorated with 120 mannose (sugar) units. This compound is able to inhibit an Ebola virus infection at sub-nanomolar concentrations [1].
Present Examples
By tweaking the electronics of aromatic compounds through the addition of unpaired or partially unpaired electrons, Yoshito Tobe and his group at Osaka University, Japan, have generated compounds with open-shell radical character [2]. These compounds have potential applications in nonlinear optics, organic electronics as semiconductors, and organic spintronics (where the electron’s spin degree of freedom plays a role). Tobe is also interested in the self-assembly of aromatic compounds on graphitic surfaces, a step on the way to functionalized graphene-based materials with tuneable properties.
Nazario Martín and Yoshito Tobe are outstanding personalities in the field of novel aromatic systems. Martín organized the International Symposium on Novel Aromatics (ISNA) in 2015 in Madrid and Tobe will be receiving the prestigious Nozoe Lecture award at the upcoming ISNA conference in Stony Brook, New York. To accompany the conference, ChemPlusChem, along with Martín and Tobe as guest editors, have put together a special issue on novel aromatics, highlighting recent developments.
What Lies Ahead in Research on Novel Aromatics?
Judging by the past, creative chemists will continue to produce new molecules and materials with distinctive properties. The key will be recognizing the “Eureka” moments, like the discovery of C60, which can dramatically change the direction of research, and also realizing the limits of individual scientists. Cooperation and collaboration will play increasingly important roles as the level of technology and cost of instrumentation increases. As Professor Riko Majima, Tohoku Imperial University, the father of modern organic chemistry in Japan, advised his students, including Tetsuo Nozoe, the founder of the ISNA symposia, “Conduct research with an infant’s curiosity and open-mindedness, and with a humble attitude.”
References
- [1] Synthesis of giant globular multivalent glycofullerenes as potent inhibitors in a model of Ebola virus infection,
Antonio Muñoz, David Sigwalt, Beatriz M. Illescas, Joanna Luczkowiak, Laura Rodríguez-Pérez, Iwona Nierengarten, Michel Holler, Jean-Serge Remy, Kevin Buffet, Stéphane P. Vincent, Javier Rojo, Rafael Delgado, Jean-François Nierengarten, and Nazario Martín,
Nat. Chem. 2016, 8, 50-57.
DOI: 10.1038/nchem.2387 - [2] Tetracyclopenta[def,jkl,pqr,vwx]tetraphenylene: A Potential Tetraradicaloid Hydrocarbon,
Shunpei Nobusue, Hirokazu Miyoshi, Akihiro Shimizu, Ichiro Hisaki, Kotaro Fukuda, Masayoshi Nakano, Yoshito Tobe,
Angew. Chem. Int. Ed. 2015, 54, 2090-2094.
DOI: 10.1002/anie.201410791
- Special Issue: Novel Aromatics: From Synthesis to Applications,
Guest Editors: Nazario Martín, Yoshito Tobe,
ChemPlusChem 2017.
Also of Interest
- Webpage Nazario Martín, Universidad Complutense de Madrid, Spain
- Webpage Yoshito Tobe, Osaka University, Japan
- New Volume of the Autograph Books of Tetsuo Nozoe,
ChemistryViews.org 2015. - Tetsuo Nozoe: Organic Chemist and Autograph Collector,
ChemViews Mag. 2012.
Tetsuo Nozoe was a pioneer in the area of non-benzoid aromatic chemistry