Vanillin is the defining scent component of the vanilla aroma. Although vanilla beans and the vanillin they contain is regarded as harmless from a health standpoint, doubts have repeatedly been raised about whether this also applies to synthetic vanillin. Despite the opposing views of the principal international health organizations, these concerns have actually found their way into a biochemistry textbook. It is high time to take up the odor trail and ferret out the true source of these doubts.
1. Vanillin
A toxicological low-blow for enthusiasts of vanilla pudding is dealt by one glance at page 86 of the textbook “Technische Biochemie” (“Technical Biochemistry”), published in 2015 [1]: “Synthetic vanillin is not something entirely free of concerns. … It has thus been classified by the ‘Advisory Body for Environmentally-Relevant Legacy Substances (BUA)’ of the German Chemical Society as belonging to the highest hazard level (+3) as carcinogenic, mutagenic, as well as a source of DNA-damage and chromosomal changes.”
A quick look at the ingredient list on a pudding container certainly leads to misgivings, since the beloved vanilla pudding generally turns out to be “original pudding with vanilla flavor”, and one searches in vain for the ingredient “vanilla”, finding instead only “vanilla flavor” and “aroma”. The European “Aroma Ordinance” [2] reveals what the true essence of this pudding is: synthetic vanillin. Since the world vanilla market amounts annually to some 15,000 t, and vanilla beans contribute only 40 t, pudding lovers must for better or worse continue to accept that synthetic vanilla lies behind the seductive scent of their dessert. And it is precisely this stuff that the textbook authors are warning us against.
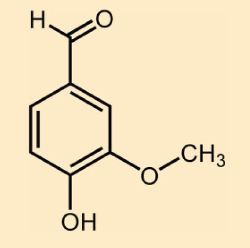
In a sense, the declared source of the doubt couldn’t be more respectable: the Gesellschaft Deutscher Chemiker (German Chemical Society, GDCh). Nevertheless, there remain nagging doubts based on one’s own acquired knowledge. Why should vanillin, i.e., 4-hydroxy-3-methoxybenzaldehyde (see Fig. 1) from a vanilla bean be harmless, but synthetic vanillin, also 4-hydroxy-3-methoxybenzaldehyde, be a health hazard?
 |
|
Figure 1. Vanillin. |
Only food-grade vanillin is considered here. Thus, there would appear to be absolutely no rational explanation. Collegial inquiries of the principal author of the textbook about possible source references, unfortunately, went unanswered.
2. Path to the Source
Our own investigations began with entering the terms “artificial vanillin” and “synthetic vanillin” into Google’s search box. Immediately below Wikipedia, which appeared as the “top hit” [3], was the “Center of Health” [Zentrum der Gesundheit], which describes itself as an uncensored and industry-independent naturopathic platform. The site’s proprietor is the Swiss firm Neosmart (!) Consulting AG. Carefully embedded product suggestions in the text and in the margins point to the purely commercial character of this platform. The Hamburg Consumer Advice Center (Hamburger Verbraucherzentrale) rates this portal as “inadequate” [4], in that its nutritional suggestions appear not to be objective, and the firm offers absolutely no information about its financial basis. Moreover, the site’s abstruse therapy suggestions are beyond all belief; a low-point is the recommended treatment for advanced prostate cancer: baking powder! [5].
This “Center of Health” comments regarding synthetic vanillin [6]: “According to the ‘Advisory Body for Environmentally-Relevant Legacy Substances (BUA)’ of the German Chemical Society, artificially prepared vanillin classes among those substances assigned to the highest hazard level (+3) as
- carcinogenic,
- mutagenic,
- capable of causing DNA damage,
- and chromosome altering.
The platform has disseminated this text through the Internet since 2010, and the authors of the 2015 edition of the textbook presumably adopted their wording from this source. In contrast to the textbook cited above, the “Center of Health” has at least provided us with a source for its information: a webpage of the Iffens Environmental Station on the Butjadigen peninsula near the UNESCO Wattenmeer World Heritage Site, not far from Bremen, Germany [7]. This facility was established over 35 years ago by the chemist Dr. Wolfgang Meiners, who characterized himself as “an Eco-animator without a financial background”. After earning his doctorate with Professor Herbert Weisz in Karlsruhe, Germany [8], Meiners took over his grandparents’ farm, subsequently extending and developing it into an ecological meeting place for young and old alike.
As early as 1984 (!), Dr. Meiners published a text under the heading “Into an eating frenzy with nature-identical flavors”, which was later put on the internet, and can still be called up from the Iffens Environmental Station webpage [9]: “The chemical substance vanillin has proved to be somewhat problematic. Today we might call it ‘environmentally relevant’, and find it in the list of 512 substances identified by the German Chemical Society’s ‘Advisory Body for Environmentally-Relevant Legacy Substances (BUA)’. Vanillin is item number 0012, and is assessed as follows: ‘Vanillin is identified as level +3 (within the range –2 to +3) as a carcinogen or mutagen in vitro or in vivo, or a source of DNA damage or chromosome alteration.'”
The list in question, containing 512 chemicals, was actually published in a report of the above-cited “Advisory Body” [10]. In order to understand how and why vanillin turned up on this list, we need to think our way back into the mindset of the 1980s.
In 1982, the German government created the aforementioned advisory body to support its own work, but intended to function under the auspices of the German Chemical Society. Its task was to select commercially available chemicals that presented a potentially hazardous threat with respect to health or the environment, and then to arrange them according to their likely threat level. Based on this priority list, the next step was to be the creation of valid substance reports regarding the most dangerous of the items, depending on the data situation, only after conducting supplemental studies.
The BUA proceeded stepwise and began by choosing the 512 most relevant of the 100,000 substances from both health and environmental standpoints (see Box 1). Given the enormous number of substances, this was possible only with the use of a very coarse screen in order to keep the challenge manageable. Vanillin was one of the 512 materials selected, since with an annual production volume at that time of 10,000 tons, it was indeed industrially significant, and was demonstrably present, for example, as a micro-impurity in the waters of North America’s Great Lakes.
|
|
|
Box 1. |
In the next step, the scientific literature for vanillin and the 511 other industrially relevant chemicals was examined in terms of eight significant environmental and health characteristics (see the left-hand column in Table 1). The results of the scientific studies were rated for their health and environmental relevance on a scale of –2 to +3. The results for vanillin are shown in Table 1. For seven of the eight environmental and health related characteristics, all the studies concurred that vanillin posed absolutely no risk whatsoever. Only the evaluation for “mutagenic or carcinogenic characteristics” [11] was assigned as “+3”, and that was enough to turn the beloved aroma substance into a threat for the first time. The source of the damning information had been found!
|
Table 1. BUA classification of the environmental- and health-relevant characteristics of vanillin [10]. |
![BUA classification of the environmental- and health-relevant characteristics of vanillin [10].](/wp-content/uploads/legacy/chem/image/2017/2017_08/vanillin_tab1_classification.jpg) |
3. A First Look at the Source
The assertion by the BUA that vanillin possesses ‘carcinogenic or mutagenic properties in vitro or in vivo, or leads to DNA damage or chromosome alteration” is devastating. Nevertheless, reference needs to be made to one detail: the BUA refers in general to vanillin, not to synthetic vanillin. If the BUA evaluation actually would apply in this way, then both synthetic vanillin and the vanilla bean, with a vanillin content of 1.5–2.5 %, would be “mutagenic or carcinogenic”. In that case, both would need to be banned. The rumor that natural vanilla is harmless but the synthetic version is dangerous is absolutely wrong, and certainly not a brainchild of the GDCh.
3.1. How Should We Evaluate the Devastating Verdict?
From the outset, it must be emphasized that the BUA assigned the highest priority to protecting people from possible damage to their genetic material caused by industrial substances. Since damage to genetic material can occur in very diverse ways, and—especially in those days—many relevant mechanisms had not been clarified in detail, the BUA decided to adopt a pragmatic approach. They took into account results of all experimental tests with respect to mutagenicity, genotoxicity, carcinogenicity, etc., so as not to overlook any potential dangers.
For this reason, they chose the following comprehensive formulation: “Carcinogens or mutagens in vitro or in vivo, or DNA damage, or potential for chromosome alteration.” The conjunction “or” was selected quite deliberately. If a substance possessed only a single one of these properties, the effect was the immediate assignment of the grade +3. Precisely this was the situation for vanillin. In fact, almost all the in vitro and in vivo tests for carcinogenicity and genotoxicity for vanillin came out negative [12], with the exception of two specific studies from a Swedish research group.
4. A Second Look at the Source
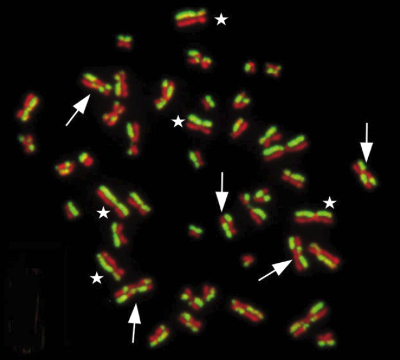
The two decisive Swedish studies examined the influence of vanillin on a special chromosomal modification: SCE, or sister chromatid exchange. This process (see Fig. 2) occurs quite naturally in cells, but it can also be accelerated chemically. As a rule, a cell, prior to division, contains two identical DNA double strands, the “sister chromatids.” In a naturally occurring process, identical DNA segments between sister chromatids exchange places, but the genetic information remains unchanged.
 |
|
Figure 2. Sister chromatid exchange. |
This exchange can be light-microscopically verified and quantified if thymidine in the DNA is replaced by the fluorescent bromodeoxyuridine (BrdU). During normal duplication, BrdU is confined to one, green-fluorescing chromatid (asterisks in Fig. 2), but in a sister chromatid exchange, fluorescing DNA segments turn up in both chromatids, forming so-called harlequin chromosomes (arrows in Fig.2). By counting harlequin chromosomes, one can quantify this process.
Since in the course of this exchange double-stranded DNA must be broken, acceleration which is induced chemically must be similar to other possible DNA modifications. Nevertheless, an in vitro SCE study involving vanillin with single-cell cultures from Chinese hamsters was negative [13], as was a corresponding study with human lymphocytes. In the latter case, only with the highest concentration (4 mmol/L) was a significant SCE increase observed, but then only if DNA damage in only a single chromatid was taken into account. In their publication, the authors summarized their results as follows [14–16]: “The prevalent food additive vanillin proved to be a strong trigger for SCEs.”
According to their own rules, and given the then current state of knowledge, the BUA had no choice but to assign vanillin a grade of +3. We repeat: the BUA was supposed to examine “only” studies, and put together only the authors’ evaluations. A technical review of their own, or true quality assessment, was not part of their assignment, and, in any case, could not have been achieved by the BUA given the massive amounts of data involved.
In the next selective step, the 512 industrially relevant chemicals were arranged in light of the eight environmental- and health-related characteristics, and the 60 most problematic were selected. Vanillin was not among them. That was no wonder, however, since among the most urgent ones to pursue at the time were candidates like benzene, benzpyrene, halogenated aromatics, and aliphatic hydrocarbons. To begin, the BUA concentrated its efforts on these 60 especially threatening substances.
In the 1980s, people’s increasing interest in questions of environmental politics encouraged corresponding initiatives in national, but also international organizations. One small step, inconspicuous at first glance, was actually quite significant. The Organization for Economic Cooperation and Development (OECD) was to have as one of its core missions the development of standards for the health evaluation of chemicals. In this context, experimental procedures and evaluations were to be prescribed in detail, and all experimental procedures to be included in the accompanying publications were to be set out in compulsory form. The goal of the OECD was to make studies themselves comparable through standardization of the various test methods. Repetition of the studies at a national level should thereby become unnecessary, helping avoid the need for redundant animal testing (see Box 2).
|
|
|
Box 2. |
As early as 1986, in the same year as the publication of the BUA, the OECD established a test procedure for “In-vitro SCE assays in mammalian cells” [17]. That was precisely the test that led, in the case of vanillin, to a +3 grade. Here, it was determined that conduct of the test and reporting of all the test parameters did not meet the OECD standards. This did not cause the study to be fundamentally unreliable, but it was criticized. The highest vanillin concentration of 4 mmol/L, equivalent to 600 mg of vanillin/L, was physiologically unrealistically high, which led to a false positive test result. Based on an overall judgment of all available data related to vanillin, the European Food Safety Authority (EFSA) therefore declared these two studies to be “not considered valid” [18].
From today’s perspective, this is not unusual, since there is no ultimate test based upon which the genotoxicity of a substance can be either unequivocally proven or excluded. More reliable are combinations of various complementary in vitro tests. Here, it has been shown specifically that tests regarding chromosomal changes frequently lead to “false positive” results [11].
Many published reports of international health organizations have shown that in the last 35 years, vanillin has been examined elaborately, thoroughly, and nearly exhaustively from a toxicological standpoint. Here is an overview:
- 1958: The American health authority U.S. Food and Drug Administration (FDA) regarded vanillin as harmless, and bestowed upon it GRAS (“generally recognized as safe”) status. This status still applies today.
- 1996: The OECD presented a 120-page comprehensive report (SIDS, Screening Information Dataset) on vanillin [12]. According to this, and on the basis of available study material, there was no health risk. Additional animal studies were regarded as unnecessary.
- 2001: The JECFA (Joint FAO/WHO Expert Committee on Food Additives) presented a report in which it was established that vanillin presented no health risks. The ADI/Acceptable Daily Intake value of 10 mg/kg body weight, established in 1967, was reaffirmed [19]. (FAO = Food and Agriculture Organization of the United Nations; WHO = World Health Organization)
- 2008: The AFC (Scientific Panel on Food Additives, Flavorings, Processing Aids and Materials in contact with Food) of the European Agency for Food Safety published a summary report over benzyl alcohols, benzaldehydes and chemically related compounds. Here, it was confirmed that there were no safety concerns whatsoever regarding the inclusion of vanillin as a flavoring agent [18].
5. Conclusion on Vanillin
For decades, the most important international health authorities have agreed—on the basis of extensive toxicological studies—that vanillin, consumed in the usual amounts, presents absolutely no health risks to humans. Pudding-lovers can, therefore, continue to enjoy their favorite food without any misgivings, because vanilla-flavored pudding is not a mutagen!
6. How Does a Rumor Long Recognized to be False Come to Appear in a Current Textbook?
It is uncontested that two scientific publications existed in 1986 in which the results of a specific test (sister chromatid exchange) pointed to a mutagenic effect of vanillin. This finding was included by the “Advisory Body for Environmentally-Relevant Legacy Substances (BUA)” of the Germany Chemical Society (GDCh) in its 1986 report [10].
 The environmental activist Dr. W. Meiners adopted the 1986 BUA data regarding vanillin, and they still appear on his webpage today as if they were engraved in stone. It is a shame that 30 years’ worth of scientific research and legislation derived from it has passed him by without a trace. The internet portal “Zentrum der Gesundheit” adopted Dr. Meiners’ text in 2010 without noting that the information had long since become obsolete. Even though this organization has tried to gird itself externally with scientific flair, it has failed here to apply appropriate caution. Numerous critical comments on the subject of artificial vanillin were either completely wiped away, or corrections were announced, but then never applied.
The environmental activist Dr. W. Meiners adopted the 1986 BUA data regarding vanillin, and they still appear on his webpage today as if they were engraved in stone. It is a shame that 30 years’ worth of scientific research and legislation derived from it has passed him by without a trace. The internet portal “Zentrum der Gesundheit” adopted Dr. Meiners’ text in 2010 without noting that the information had long since become obsolete. Even though this organization has tried to gird itself externally with scientific flair, it has failed here to apply appropriate caution. Numerous critical comments on the subject of artificial vanillin were either completely wiped away, or corrections were announced, but then never applied.
Thus, Nuong Trang inquired in 2011 “if true vanillin can also have the carcinogenic effects of the synthetic material.” The absurd reply: “Natural vanilla contains vanillin along with hundreds of other substances. In other words, vanillin is present here to a tiny extent, and together with all the other natural components of vanilla, so that one cannot assume that it has carcinogenic effects. However, if vanillin is isolated and used in concentrated form, then there could be adverse consequences” [6]. The site answer critique in 2011 with: “On this occasion, we would gladly seize your suggestion, and before the next updating of the text we will ask the German Chemical Society for a clarification of the situation, and incorporate it into the updated article.” In 2014, it stated: “Many thanks for your commentary and your remarks. Our vanillin article will soon be correspondingly revised, corrected, and updated.” as well as “We will be glad to examine both the source as well as the article, and revise it as appropriate. Thank you for your suggestion.” No inquiry was made at the GDCh, nor has anything been changed in the meantime. [6]
The authors of the textbook “Technische Biochemie” must finally be charged with uncritically adopting in an introductory textbook text modules from a dubious source.
Acknowledgements
An inquiry by the scientific editor Peter Kötzing, Nuremberg, Germany, through the GDCh, elicited the background research cited here. Dr. Renate Hoer of the GDCh, Frankfurt, Germany, helped with initial suggestions, and Dr. Heidrun Greim, Munich, Germany, provided important suggestions regarding the operating procedures of the BUA and helped in ferreting out old documents. Dr. Wolfgang Meiners, Butjadingen, Germany, specified more precisely his source materials. I wish to thank Dr. S. Streller of the Free University of Berlin, Germany, for her creative content suggestions and critical review of the manuscript, and Dr. P Winchester, Free University Berlin, for her linguistic help.
References
[1] O. Kayser, N. Averesch, Technische Biochemie (in German), Springer Fachmedien, Wiesbaden, Germany, 2015, 86. ISBN: 978-3-658-05548-6
[2] European Flavor Ordinance (in German), December 16, 2008.
[3] www.google.de (accessed 2016)
[4] Verbaucherzentrale Hamburg, Wer und was versteckt sich hinter Ernährungsportalen? (in German), www.vzhh.de, 2013. (accessed 2016)
[5] Zentrum der Gesundheit, Mit Backpulver (Natron) von Krebs geheilt (in German), www.zentrum-der-gesundheit.de. (accessed 2016)
[6] Zentrum der Gesundheit, Vanillin – Synthetische Vanille (in German), www.zentrum-der-gesundheit.de. (accessed 2016)
[7] Website of the Umweltstation Iffens (in German), www.umweltstation-iffens.de. (accessed 2016)
[8] H. Weisz, W. Meiners, Double indication in catalytic—kinetic analysis; Determination of iron(III), cyanide and molybdenum(VI), Anal. Chim. Acta 1977, 90, 71. https://doi.org/10.1016/S0003-2670(01)82295-9
[9] Naturidentisch in den Fressrausch (in German), www.umweltstation-iffens.de. (accessed 2016)
[10] Beratergremium für umweltrelevante Altstoffe, Gesellschaft Deutscher Chemiker (Ed.) Umweltrelevante Alte Stoffe, Auswahlkriterien und Stoffliste (in German), VCH, Weinheim, Germany, 1986. ISBN: 3527266178
[11] H.-W. Vohr (Ed.), Toxikologie: Grundlagen der Toxikologie, Band 1 (in German), Wiley-VCH, Weinheim, Germany, 2010. ISBN: 9783527323197
[12] Vanillin: SIDs Initial Assessment Profile, UNEP Publications. (accessed 2016)
[13] A. Kasamati et al., Genotoxicity of flavoring agents, Mutat. Res. 1982, 105, 387. https://doi.org/10.1016/0165-7992(82)90181-6
[14] T. Jansson, The frequency of sister chromatid exchanges in human lymphocytes treated with ethanol and acetaldehyde, Hereditas 1982, 97, 301. https://doi.org/10.1111/j.1601-5223.1982.tb00883.x
[15] M. Curvall, T. Jansson et al., In vitro studies of biological effects of cigarette smoke condensate: II. Induction of sister-chromatid exchanges in human lymphocytes by weakly acidic, semivolatile constituents, Mutat. Res. 1986, 169, 129. https://doi.org/10.1016/0165-1218(86)90092-3
[16] T. Jansson. L. Zech, Effects of vanillin on sister-chromatid exchanges and chromosome aberrations in human lymphocytes, Mutat. Res. 1987, 190, 221. https://doi.org/10.1016/0165-7992(87)90033-9
[17] OECD Test Guidelines for Testing of Chemicals: Introduction to the OECD Guidelines on Genetic Toxicology Testing, www.oecd.org. (accessed 2016)
[18] European Food Safety Authority (EFSA), Flavouring Group Evaluation 52, EFSA J. 2008, 6, 637. https://doi.org/10.2903/j.efsa.2008.637
[19] Fifty-seventh report of the Joint FAO/WHO Expert Committee on Food Additives, who.int, 2002.(accessed 2016)
The article has been published in German as:
- Ist Pudding mit Vanille-Geschmack mutagen?,
Klaus Roth,
Chem. Unserer Zeit 2016, 50, 226–232.
DOI: 10.1002/ciuz.201600758
and was translated by W. E. Russey.
See all articles by Klaus Roth





Thank you for this very informative article which settled a marital dispute on the possible effects of vanillin consumption by humans. I was especially gratified that the sources of the controversy were so thoroughly explored and explained in terms accessible to non-chemists. M Lutz, PhD, MPH