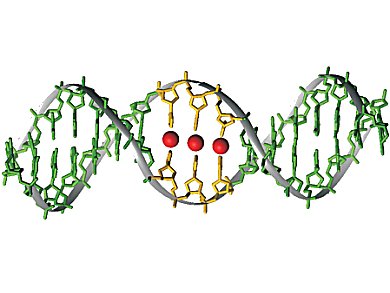
The highly versatile biomacromolecule DNA is increasingly being applied as a supramolecular scaffold for functional molecules. Over the last decade, a new twist has been added to this approach: The generation of so-called metal-mediated base pairs elegantly allows the site-specific introduction of metal-based functionality.
In their review article, Jens Müller and Philipp Scharf, University of Münster, Germany, provide an introduction into this topical and highly interdisciplinary area of research. They have compiled the results of scientists around the world and present an overview of metal-mediated base pairs devised so far as well as of the elucidation of their chemical and physical properties.
In metal-mediated base pairs, the hydrogen bonds present in natural base pairs are formally replaced by coordinative bonds to metal ions located along the helical axis. They can be made by introducing artificial ligand-based nucleosides into the nucleic acid sequence. In the presence of suitable transition metal ions, these metal ions will site-specifically bind to the nucleic acid. As a result, DNA can be used as a supramolecular scaffold for metal-based functionality, adding new physical properties to this biomacromolecule.
Metal-mediated base pairs are important because they represent an elegant approach for expanding the genetic code. Moreover, they can be applied in highly specific metal ions sensors for aqueous environments. The highly versatile applicability in various areas will lead to many more applications in the future. The latest developments in this respect are highlighted in the review article.
- Nucleic Acids with Metal-Mediated Base Pairs and Their Application,
Philipp Scharf and Jens Müller,
ChemPlusChem 2013.
DOI: 10.1002/cplu.201200256