New Type of Magnetic Stir Bars
Anyone who has ever worked in a laboratory has seen them: magnetic stirrers that rotate magnetic stir bars in liquids to mix them. The stir bars come in many different forms—now including nanometer-sized. In the journal Angewandte Chemie, researchers from Singapore have now introduced chains made of 40 nm iron oxide particles that act as the world’s smallest magnetic stir bars, effectively stirring picoliter-sized drops of emulsion with a commercial magnetic stirrer.
Effective stirring is essential in chemical and biological experiments. This is usually achieved with magnetic stirrers and stir bars. However, this does not work in the tiny channels and droplets used in lab-on-a-chip applications and for microliter-scale experiments in the biosciences. Inexpensive stir bars that are small enough but still able to absorb external magnetic energy and efficiently translate it to stir tiny volumes are thus high on the wish list.
The problem lies in the tiny size of previous micrometer-sized stir bars: They are too big to remain suspended because they are pulled to the bottom of the vessel by both gravity and magnetic attraction. At the same time, they are too small to completely stir the solution when they are on the bottom, which works for macroscopic stir bars. The majority of the liquid remains unmixed.
Stir Bars So Small they Remain in Suspension
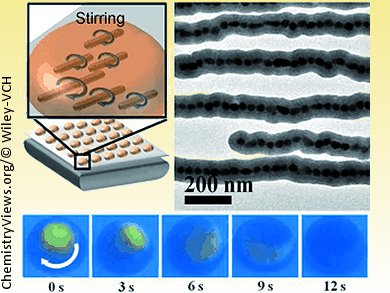
A team led by Hongyu Chen, Nanyang Technological University, Singapore, has now found a solution to this problem: tiny silicon dioxide coated rods made of lined-up iron oxide nanoparticles. They are even easy to make. Magnetic iron oxide particles with diameters of 40 nm are stabilized with oleic acid, modified with citric acid to make them water soluble, and dispersed in a water/propanol mixture. After addition of an organosilicon compound and ammonia, the reaction vessel is simply left to stand near a magnet overnight. The stir bars can then simply be collected by centrifugation.
The thickness of the silicon layer can be controlled, allowing for the production of stir bars with diameters ranging from 75 nm to 1.4 µm. Their length can reach up to 17 µm. The bars are thus so small that they remain suspended in solution. Addition of a large number of stir bars ensures that all of the liquid is stirred. In the magnetic field of a conventional magnetic stir plate, the individual stir bars move independently. It is thus possible to thoroughly mix droplets of just a few picoliters.
The nanoscale stir bars can be easily removed by adding the droplets on top of a strong magnet wrapped in a layer of plastic film. The magnetic field gradually pulls the stir bars to the bottom of the droplets, and the droplets can then simply be picked up with a pipette.
- Stirring in Suspension: Nanometer-Sized Magnetic Stir Bars,
Hongyu Chen,
Angew. Chem. Int. Ed. 2013.
DOI: 10.1002/anie.201303249