There’s a moment in Disney’s animated epic “Frozen” where the heroine uses her “icy powers” to produce snow, and lots of it. If only it was this simple in real life! To safeguard against nature’s unpredictability, snowmakers continue to find ways of manufacturing snow on a large scale. And the heat is on: Europe felt its warmest winter on record in 2015–16; Switzerland is entering its third consecutive Christmas season with little or no snowfall, whereas in the southern French Alps snowfall is down as much as 80 % [1].
Worryingly, winter 2015 in California, US, produced the worst snowpack in 500 years [2]. Ski resorts worldwide increasingly depend on some form of artificial (machine-made) snow. But how was it first made? How has snowmaking improved over the years? And, considering the rising temperatures and the financial and environmental costs of snowmaking, does it have a future?
1. What is Snow?
 Snow crystals form when water vapor in the atmosphere condenses directly into ice in the form of a crystal-lattice structure [3]. This process usually begins when the wind causes warm, moist air to collide with another mass of air. The collision pushes the warm air mass upward and the air cools as it rises. Once the air cools sufficiently, some of the water vapor it carries condenses into water droplets. Each droplet requires a nucleus on which to condense; particles of dust in the air serve this purpose.
Snow crystals form when water vapor in the atmosphere condenses directly into ice in the form of a crystal-lattice structure [3]. This process usually begins when the wind causes warm, moist air to collide with another mass of air. The collision pushes the warm air mass upward and the air cools as it rises. Once the air cools sufficiently, some of the water vapor it carries condenses into water droplets. Each droplet requires a nucleus on which to condense; particles of dust in the air serve this purpose.
If the newly formed clouds continue to cool and the temperature drops below 0 °C, water droplets do not freeze immediately. Instead, they remain liquid in what is called a supercooled state. Pure water droplets can be supercooled to nearly –40 °C before they freeze. But dust-containing droplets begin to freeze at around –6 °C, because dust provides a solid surface or “ice nucleus” that kickstarts the freezing process. Once an individual droplet freezes, water vapor condenses onto its surface and freezes and the crystal begins to grow and develop. The snow crystal becomes heavier as it grows, until it falls under gravity. The liquid droplets in the cloud that remain unfrozen slowly evaporate, supplying the air with the water vapor that creates other ice crystals. Therefore, snow crystals are made from water vapor, not liquid water.
2. The First Lab-Grown Snowflake
It was a Japanese physicist, Ukichiro Nakaya, who synthesized the first snowflake in the lab. Upon his arrival at Hokkaido University in the 1930s, Nakaya found that the only thing in abundance was natural snow. Nakaya was clearly passionate about snow crystals, he called them “letters sent from heaven” [4]. However, the recreation of these “heaven-sent” crystals was more difficult than Nakaya anticipated.
In 1936, Nakaya had a breakthrough when he found a snow crystal on the tip of a hair of a rabbit fur coat in the lab. Having found a suitable ice nucleus, Nakaya then built a chamber with adjustable temperature, air pressure, and humidity to mimic cloud-like conditions. In a detailed series of experiments circulating water vapor in this cooled chamber, Nakaya grew many types of snow crystals on the tip of a rabbit’s hair [5,6]. Nakaya was the first to observe that the temperature and humidity of the air determined the basic shape of an ice crystal.
3. Machine-Made Snow
3.1 Early Snowmaking Machines
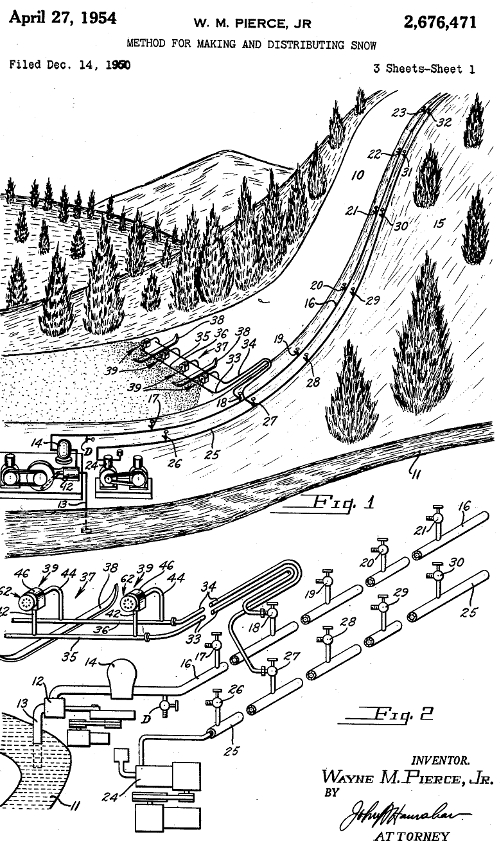 A Canadian researcher, Ray Ringer, discovered the first snowmaking machine accidentally in the 1940s. Ringer was investigating the formation of rime (a form of ice) on jet engines. Ringer and colleagues sprayed water into a chilled wind tunnel in front of a jet engine, but instead of producing a layer of ice as Ringer had anticipated, the cooled water froze mid-air and snow crystals sprayed from the back end. The researchers failed to commercialise the results, but their findings inspired Wayne Pierce and colleagues (Tey Manufacturing, Connecticut, US) to design a similar device based on Ringer’s findings (see patent pictured on the right). The first DIY snow gun was born out of a paint spray compressor, nozzle and a garden hose [7].
A Canadian researcher, Ray Ringer, discovered the first snowmaking machine accidentally in the 1940s. Ringer was investigating the formation of rime (a form of ice) on jet engines. Ringer and colleagues sprayed water into a chilled wind tunnel in front of a jet engine, but instead of producing a layer of ice as Ringer had anticipated, the cooled water froze mid-air and snow crystals sprayed from the back end. The researchers failed to commercialise the results, but their findings inspired Wayne Pierce and colleagues (Tey Manufacturing, Connecticut, US) to design a similar device based on Ringer’s findings (see patent pictured on the right). The first DIY snow gun was born out of a paint spray compressor, nozzle and a garden hose [7].
However, the early Tey machines were very noisy and expensive to run. In addition, water would freeze in the pipes if pumped too slowly. In 1961, Alden Hanson was issued a patent for the use of a fan, particulate water and a nucleating agent (dirt particles), which was quieter, cheaper to run, and produced a lot more snow [8]. The Hanson patent is considered the pioneer patent for all fan snowmaking machines.
3.2 Current Snowmaking Machines
There are two main types of snowmachines commercially available: fan guns and snow lances [9]. Both use nucleating agents to kickstart the process. Machine-made snow gives tiny, hollow spheres that are up to five times as dense as fresh snow. As a result, machine-made snow is more durable and can be heavily groomed before it loses its properties. However, this type of snow is not always better for skiing.
3.1.1 Snow Lances
A snow lance is a long aluminum tube that has air and water nozzles at the tip. As the air and water mix, the compressed air expands and cools, creating ice nuclei for the crystallization of water. Snow lancers are cheaper than fan guns, but have limited throw and are very sensitive to wind.
 3.1.2. Fan Guns
3.1.2. Fan Guns
Instead of a compressor, fan guns (pictured left) use a high-powered electrical fan that can fire particles up to 50 m away. The longer “hang time” allows the formation of larger snowflakes. Fan guns are known for long snow projection and throw in all weather conditions, low wind sensitivity, and good overall performance.
3.3 Snowmaking Conditions
Snowmaking machines are highly sensitive to changes in temperature and humidity, so snowmakers measure the “wet bulb” temperature, which defines the coldest condition a water droplet can obtain, taking into account both the ambient temperature and relative humidity. The name is derived from the technique of putting a wet cloth over the bulb of a mercury thermometer and then blowing air over the cloth until the water evaporates. This reading is typically colder than the “dry bulb” temperature, which is the normal temperature on a thermometer. The colder and drier the conditions, the more effective the snowmaking process is.
Advances in technology mean that snowmaking equipment can now be automated from a central computer or even a smartphone. Automation promises improved snow quality and efficiency, simplifying the constant machine adjustments to changing conditions, which results in reduced labor costs [10].
Because snowmaking machines use vast quantities of water, ideally, the source should be surface water stored in cooled snowmaking reservoirs, which fill up naturally from rainfall and snowmelt. At the end of the season, the water returns to the pond.
3.4 Snowmaking Additives
3.4.1 Ice-Nucleating Proteins (INPs)
At warmer temperatures (above –7 °C), ice will not form spontaneously. Instead, snowmakers often use additives to initiate the nucleation process. One of the most active nucleators is a “frosty” bacterium that floats around in the atmosphere.
In 1975, Steve Lindow, a graduate student in plant pathology at the University of California, Berkeley, USA, found that Pseudomonas syringae bacteria produce proteins that cause water to freeze at fairly high temperatures (–4 °C to –2 °C) [11]. In nature, ice-forming bacteria like P. syringae rely on a unique ice-nucleating protein (INP) that is present on their surfaces. The structure of the INP mimics the surface of an ice crystal, acting as a template that forces neighbouring water molecules into a pattern, similar to that of an ice crystal lattice. This process lowers the amount of energy needed for ice crystals to start growing.
The findings were commercialized in 1987, resulting in a product named Snowmax. Bacteria are grown in large-scale fermentation, then pelleted, freeze-dried, and finally irradiated [12]. The product is then hydrated on site and added into a water supply. According to the manufacturers, Snowmax International (Colorado, USA), snowmakers can achieve up to 40 % more snow by spraying the treated water high over the snow area. Whereas other high-temperature nucleators can only be used up to –6.5 °C, their product works at –0.6 °C and in lower humidity [13].
However, the use of Snomax is not without controversy; it appears that certain fungicidal proteins in the bacteria retain their activity even after the dehydration and irradiation treatment. Concerns over Snowmax’s fungicidal effect on the environment have led to calls for further investigation over its environmental impact. Scientists are now looking at ways of making a Snomax-like additive without the fungicidal effect [14].
3.4.2 Non-Nucleating Additives
Not all additives work to initiate nucleation. The organosilicone surfactant “Drift” made by Aquatrols, New Jersey, US, is a surfactant that is added to water. Drift breaks down the cohesive tendencies of water and allows it to form and flow as a mass of flatter or thinner droplets [15]. When the guns spray the pre-treated water, the droplets disperse into a finer mist because of the greater surface area of the droplets. Furthermore, the energy holding the water molecules together is lowered by the surfactant. This means that Drift can help convert water to ice much more rapidly and thoroughly as it is blown into the cold air. The more particles form snow crystals in the air, the drier the snow is on the slope. According to the manufacturers, a higher ratio of water to air can be used, reducing energy costs and increasing snow volumes.
4. Sustainable Snowmaking
4.1 Vacuum Snowmakers
When the weather is too warm, weather-dependent snowmaking technologies are often redundant. However, a vacuum-based technology, the Vacuum Ice Maker (VIM) (IDE Technologies, Israel), makes snow regardless of air temperature [16].
By exposing water to a high vacuum in a freezer, the vacuum forces part of the water to evaporate, while the remaining water freezes. The water vapor is continuously evacuated from the freezer, compressed and fed into condenser by a centrifugal compressor. A standard water cooler chills the water at 5 °C, which condenses the vapor. The mixture is pumped from the snow generator to a snow concentrator, which separates the water from the snow crystals. Although the density of this snow (600–700 kg/m3) is almost double that of traditional machine-made snow, the manufacturers claim that it uses 80 % less power than other weather-independent snowmaking technologies. Resorts in Zermatt, Switzerland, and Pitztal, Austria, use this type of snowmaker to extend their ski seasons.
4.2 Cloud-Seeding Efforts
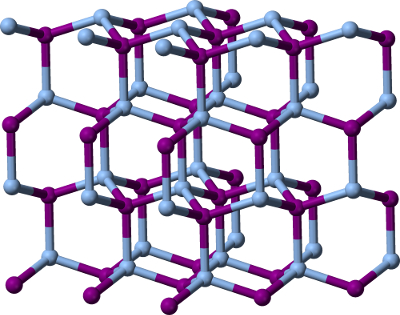 Weather modification may seem like a drastic measure, but scientists have been trying to make weather “on-demand” since the 1940s. In 1946, General Electric Laboratories (New York, US) scientist, Vincent Schaefer, successfully used dry ice to induce precipitation in a cloud over a mountain range in upstate New York, US. The snow melted before it hit the slopes below, but it was documented as the first artificially produced snowfall [17].
Weather modification may seem like a drastic measure, but scientists have been trying to make weather “on-demand” since the 1940s. In 1946, General Electric Laboratories (New York, US) scientist, Vincent Schaefer, successfully used dry ice to induce precipitation in a cloud over a mountain range in upstate New York, US. The snow melted before it hit the slopes below, but it was documented as the first artificially produced snowfall [17].
To investigate whether the technique can increase snowfall, meteorologists at the National Centre for Atmospheric Research (NCAR) in Colorado, US, sprayed silver iodide particles into storm clouds over mountains in Wyoming during the winters of 2008–2014 [18]. The crystalline structure of AgI (pictured on the right) is nearly identical to that of ice. Therefore, supercooled liquid water in the cloud freezes on the AgI particles, initiating nucleation. Results suggest that seeding could boost precipitation by 5–15 % in optimal conditions. Although researchers caution that these are best-case results, as the ideal conditions required only occur in about a third of cloud events.
4.3 Artificial Snow Cloud
If real clouds fail to produce snow, researchers from the University of Natural Resources and Life Sciences in Vienna and the Vienna University of Technology in Austria may have a solution: An artificial snow cloud “dendrite generator”.
Inside a real cloud, a developing snow crystal experiences changing temperatures and humidity levels. The device mimics cloud conditions by mixing a flow of humid and a flow of cold air in a chamber, which forms an atmosphere super-saturated with water. The liquid is supercooled at low temperatures (below –5 °C), and a short pulse of pressurized air triggers nucleation [19]. The growing ice crystals and snowflakes are suspended by carrier airflow, which is sprayed through an opening in the chamber.
The 11.5 m bright-green cloud chamber is currently being tested on the slopes of the Obergurgl-Hochgurgl resort in Tyrol, Austria [20]. Whereas snow cannons use 1 cm3 of water to produce about 2 cm3 of snow, the artificial cloud can produce up to 15 cm3 of powder snow from only 1 cm3 of water. The Obergurgl-Hochgurgl ski lift company spends € 3 million a year to cover 95 % of its 110 km slopes with machine-made snow. Researchers believe that the artificial cloud will result in a dramatic reduction in energy and costs. However, the conditions have to be ideally colder than –5 °C, and powder (dendrite) snow will only start forming at –10 °C [21]. Other factors also affect the amount of snow produced, such as wind and temperature.
If lower cost cloud-seeding efforts or artificial cloud technologies continue to show some benefit, they could have a future in “green” snowmaking. As a short-term solution to unseasonably warm weather, ski resorts are likely to increase their output of machine-made snow, but even this “insurance policy” becomes redundant once the temperature increases above freezing. For now, snowmaking continues to be ruled by the weather, but rising temperatures will no doubt force investment towards new weather-independent snowmaking technologies.
References
[1] Where’s the snow? Alpine ski resorts count cost of a warm Christmas, euronews.com 2015. (accessed January 12, 2016)
[2] S. Belmecheri et al., Nature Clim. Change 2016, 6, 2. DOI: 10.1038/nclimate2809
[3] K. Libbrecht, American Scientist 2007, 95, 1, 52. DOI: 10.1511/2007.63.52
[4] U. Nakaya, Snow Crystals, Natural and Artificial. Harvard University Press, Cambridge, 1954. ISBN: 978-0-674-81151-5
[5] U. Nakaya et al., J. Faculty Sci., Hokkaido Imp. Univ. 1938, Ser. II, 1. Link
[6] U. Nakaya etal., J. Faculty Sci., Hokkaido Imp. Univ. 1938, Ser. II, 2. Link
[7] W. Pierce, US Patent 2676471 A, issued April 1954.
[8] A. Hanson, US Patent 2968164, issued January 1961.
[9] Snowmaking Basics, snowmakers.com. (accessed 17 Jan 2016)
[10] E. Spence, Fake Snow, Real Money: The High-Tech Fight to Save California Skiing, bloomberg.com. (accessed 17 Jan 2016)
[11] D. C. Arny et al., Nature 1976, 262, 282. DOI: 10.1038/262282a0
[12] A. Margaritis, A. S. Bassi, Crit. Rev. Biotechnol. 1991, 11, 277. DOI: 10.3109/07388559109069185
[13] Effects of Snomax, snomax.com. (accessed January 19, 2016)
[14] J. Y. Takemoto, US Patent 20060248793 A1, issued November 2006.
[15] What is Drift, driftsnow.cz. (accessed January 20, 2016)
[16] All Weather Snow Making, ide-snowmaker.com. (accessed January 21, 2016)
[17] Langmuir Laboratory for Atmospheric Research, Storms Above the Desert, langmuir.nmt.edu. (accessed January 21, 2016)
[18] A. Witze, Major cloud-seeding test gives mixed results, Nature News 2014. (accessed January 19, 2016).
[19] A. Valenta, Dendrite Generator – Apparatus for Artificial Snow Production, tuwien.ac.at. (accessed January 20, 2016)
[20] World Premiere in Obergurgl: Artificial Cloud Produces Snow at the Touch of a Button, obergurgl.com. (accessed January 18, 2016)
[21] Deutsche Welle, Artificial Snow Goes Green, dw.com 2015. (accessed January 16, 2016)





skied on snowmax first time in WV, USA. It was fine during cold temperatures, seemed to be normal like machine made snow… However, when the temperature warmed up which makes the snow soft and granulated, the skis would slow down as if the snow was no longer slippery. It was like hitting wet grass, a very noticeable slow down. I have been skiing on all snow types for 40 years.