Sarvesh Kumar Srivastava and colleagues from IFW Dresden, Germany, developed catalytic micromotors that use water pollutants for fuel. They require no pH or temperature manipulation, they completely degrade the target compounds, and they can be recovered using a magnet.
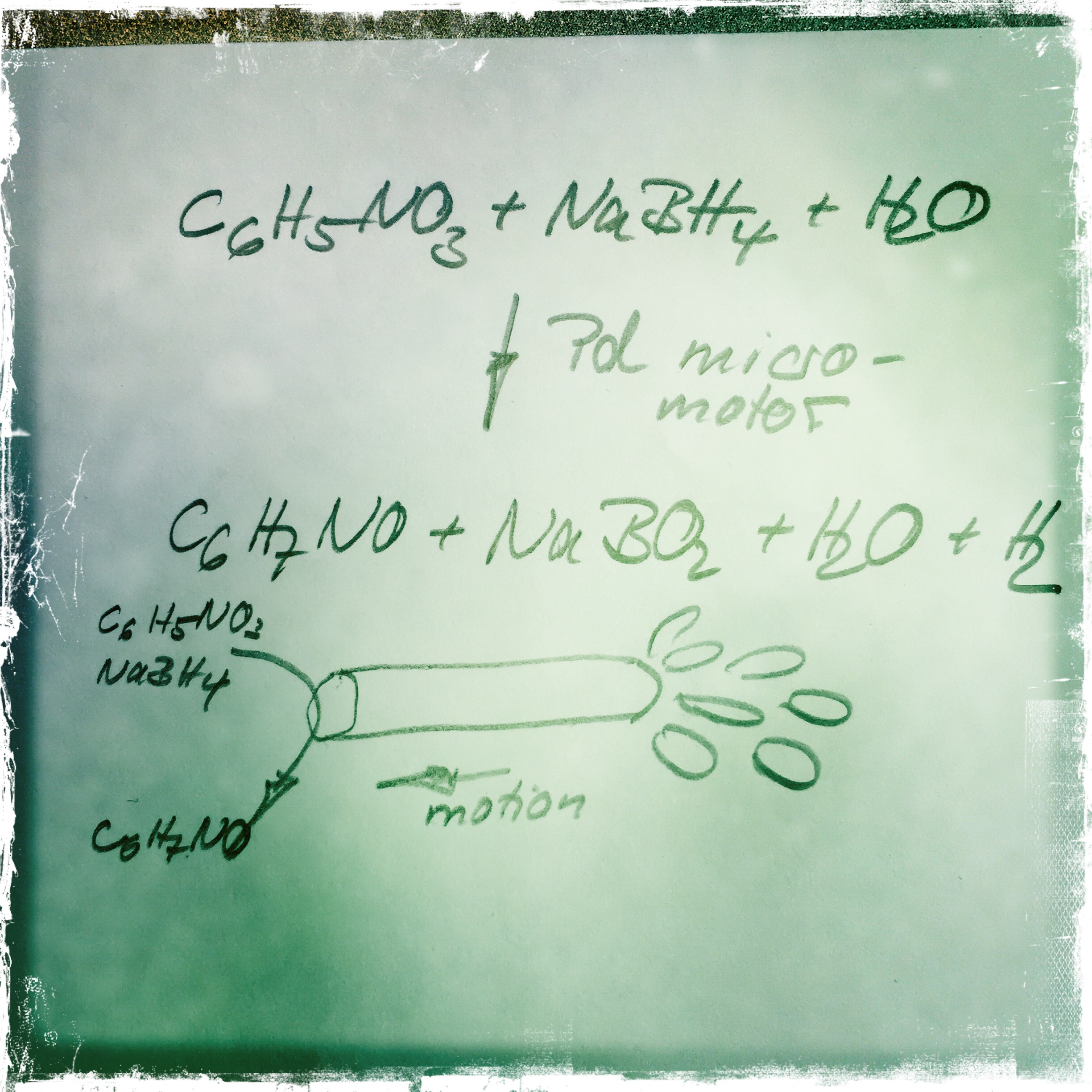
The motors consist of thin Ti/Fe/Cr films rolled into tubes with catalytically active Pd particles coating the inside surfaces. The researchers tested the motors on 4-nitrophenol and sodium borohydride, compounds used in the leather, textile, and pulp industries. When these two pollutants react, they form yellow 4-nitrophenolate ions. The disappearance of the yellow color provided a convenient way to track the progress of the cleanup. The catalyst-driven reaction generated bubbles that propelled the tubes through the water as they converted the pollutants into nontoxic 4-aminophenol and borate species.
Because water flowed through the micromotors, they cleaned up pollutants 10 times faster than their stationary counterparts. This method requires no external fuel, surfactants, or light sources, and it does not harm microorganisms in the water. The micromotors degraded more than half of the 4-nitrophenol within the first minute and 99 % after 10 minutes.
- Wastewater Mediated Activation of Micromotors for Efficient Water Cleaning,
Sarvesh Kumar Srivastava, Maria Guix, Oliver G. Schmidt,
Nano Lett. 2016.
DOI: 10.1021/acs.nanolett.5b0503