Recovering Precious Metals from Electronics
“Urban mining”, the recycling of precious metals from electronic gadgets, becomes ever more important. However, processes that are both efficient and environmentally benign are still scarce. Timo Repo, University of Helsinki, Finland, and colleagues have looked deeper into gold dissolution, in particular, how organic thiol-containing compounds help dissolve elemental gold. They propose a selective, fast, and convenient thiol-assisted gold leaching processes.
The traditional way of recycling gold “waste” is melting: dental gold and jewelry can be recycled close to 100 %. Recycling of precious metals in smartphones, computers, and other electronic gadgets is much harder, and the recovery quote is still low. Despite the abundance of precious metals in electronic devices, their relative content is still too low to allow for really economical urban mining.
Complexation of Gold in Organic Solutions
The traditional mining method for gold is hydrometallurgical cyanide leaching, which produces a vast amount of hazardous waste while being relatively unselective. More recent concepts rely on the complexation of gold in organic solutions. The metal can form soluble complexes with sulfur-containing reagents. However, the processes must be feasible on a large scale and avoid toxic or hazardous compounds.
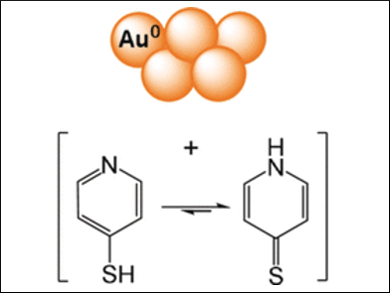
The team proposes an efficient gold recovery method from electronic waste with pyridinethiols and hydrogen peroxide as reagents, dimethyl formamide as an organic solvent, and, optionally, elemental sulfur to reduce the reagent load. Pyridinethiol (pictured) is a nitrogen-containing aromatic ring with a thiol group.
The reagent not only binds elemental gold to form soluble complexes, but the complex also has a favorable linear structure formed by two pyridinethiol molecules on either side of the gold atom. Upon oxidation, it transforms into a stable cationic gold-containing product in organic solution. This complex formation with two ligands is a specialty of gold and favors the dissolution and oxidation. Accordingly, the researchers reported nearly quantitative dissolution of gold from powder, film, or electronic boards after 20 min extraction time.
Selective Dissolution of Elemental Gold
But how can gold dissolution be distinguished from that of other precious metals? In contrast to gold having a one-electron oxidation, platinum and palladium require two-electron oxidations and, thus, are not accessible with this method. In contrast, both copper and silver form complexes with pyridinethiols, although not as effective as gold. Therefore, before dissolving the gold from the “gold finger” region in a printed circuit board, the scientists first extracted copper and silver with ammonia and sulfate-containing solutions, which are established methods.
Looking into the exact mechanism of thiol-assisted gold dissolution, the scientists discovered a surprisingly high variety of sulfur-containing side products. Some of them seemed to be crucial for proceeding the oxidation reaction, for example S8, a common form of elemental sulfur. This also proved to be an asset: By adding external S8, the ligand load could be reduced. The developed extraction method could be a basis for more efficient urban mining.
- Pyridinethiol-Assisted Dissolution of Elemental Gold in Organic Solutions,
Minna Räisänen, Eeva Heliövaara, Feda’a Al-Qaisi, Mikko Muuronen, Aleksi Eronen, Henri Liljeqvist, Martin Nieger, Marianna Kemell, Karina Moslova, Jani Hämäläinen, Kalle Lagerblom, Timo Repo,
Angew. Chem. Int. Ed. 2018.
https://doi.org/10.1002/anie.201810447