Asparagus is highly prized in culinary circles. However, enjoying asparagus has an unpleasant side-effect that sets it apart from other vegetables, but is seldom talked about: It makes urine stink. This time, we’ll look at how asparagus changes when it is cooked.
5. The Odor of Cooked Asparagus
The first step to enjoying asparagus is the cooking process, which amounts to a lot more than simple physical warming [20,21]. The breaking of weak hydrogen bonds and resulting softening of hard and tough superstructures made of proteins and polysaccharides is just one part of the cooking process. Heating to 100 °C involves the components of this vegetable in myriad chemical reactions both alongside and with each other, making the cooking pot a place where some of the most complex and confusing chemistry on the planet occurs.
While cooking, the reactive sulfur-containing components of raw asparagus at the focus of this article undergo massive degradation reactions (see Fig. 7). We will concentrate on the search for suitable starting materials for the sulfur-containing compounds in urine.
.jpg) |
|
Figure 7. Cooking asparagus. Three chemically independent processes contribute to the odor: degradation of reactive sulfur compounds (top), the oxidative degradation of unsaturated fatty acids (middle), and the Maillard reaction of (also sulfur-containing) amino acids with reduced sugars. |
Of the sulfur-containing components in asparagus, cooking primarily breaks down S-methylmethionine (11) to form dimethylsulfide (4) [22, 23]. With a boiling point of 37 °C, this compound evaporates in the cooking process and is a primary component of the scent of cooked asparagus. Dimethylsufide (4) is formed during the cooking process of nearly all vegetables. The odor is particularly intense for our noses when we cook cabbage and legumes.
The thermal decomposition products of asparagusic acid (12) include 3H-1,2-dithiol (15) and 1,2,3-trithia-5-carboxylic acid (16). Compound 15 is a particularly significant component of the characteristic smell of cooked asparagus [24]. The decomposition processes have a positive effect on the flavor, since asparagusic acid gives raw asparagus a strongly astringent flavor [24].
The cooking process also involves hundreds of other chemical reaction cascades that are not related to sulfur-containing compounds. Of these, the oxidative splitting of unsaturated fatty acids and the reaction of amino acids with reducing sugars (Maillard reaction) contribute strongly to the flavor (see Fig. 7).
6. Eating Asparagus and the Fragrant Finish
The enjoyment of asparagus begins as the smell of cooked asparagus rises into the nose. This scent is composed of an entire bouquet of volatile compounds formed by various reaction pathways during the cooking process (see Fig. 7) [24]. While we usually take up only the tiniest amounts of these substances through our noses, larger amounts (10 mg or so) of sulfur-containing components enter our metabolism when we eat, depending on our appetite. Which of these could be precursors of the urine odor components is unclear, particularly since the two most promising candidates, S-methylmethionine (11) and asparagusic acid (12), are mostly broken down by cooking.
An important clue is provided by Roland Tressl and coworkers [20], according to whom the concentration of methyl asparagusinate (14) in raw asparagus is significantly higher than that of free asparagusic acid (12). The methylester 14 withstands cooking without damage, in contrast to the free acid 12.
At this point we should remember the observation of John Arbuthnot (1667–1735), the Scottish mathematician and physician to Queen Anne: “Asparagus affects the urine a fetid smell (particularly if it was picked while still white) and, therefore, has been suspected by some physicians as not friendly to the kidneys. When it grows older and begins to branch out, it loses this property and is no longer considered as such.” (also see Fig. 1 in Part 1)
 The observation that only the white asparagus spears grown underground lead (see Part 1) to the characteristic urine odor is supported by more recent chemical experiments. These have shown that the concentrations of asparagusic acid and its derivatives decrease sharply after the spears break ground and photosynthesis begins [25]. This link between an observation from the 18th century and modern chemical investigations points to asparagusic acid derivatives as the source of the asparagus urine odor.
The observation that only the white asparagus spears grown underground lead (see Part 1) to the characteristic urine odor is supported by more recent chemical experiments. These have shown that the concentrations of asparagusic acid and its derivatives decrease sharply after the spears break ground and photosynthesis begins [25]. This link between an observation from the 18th century and modern chemical investigations points to asparagusic acid derivatives as the source of the asparagus urine odor.
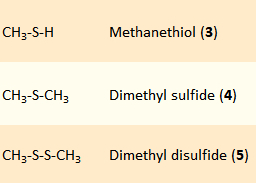
It is unclear where in our body (in which organ) and how (through enzymatic reactions or thermal decomposition) the volatile scent components 3–5 (pictured on the right) are produced from methyl asparagusinate. We are not even sure if we, not symbiotic organisms in our digestive tract, are actually responsible. We are sure of one thing, though: we pee out the stinking trio 3–5.
References
[20] R. Tressl et al., Formation of flavor components in asparagus. 2. Formation of flavor components in cooked asparagus, J. Agric. Food Chem. 1977, 25, 459–463. https://doi.org/10.1021/jf60211a048
[21] D. Ulrich et al., Contribution of volatile compounds to the flavor of cooked asparagus, Eur. Food Res. Technol. 2001, 213, 200–204. https://doi.org/10.1007/s002170100349
[22] R. Tressl et al., Formation of flavor components in asparagus. 1. Biosynthesis of sulfur-containing acids in asparagus, J. Agric. Food Chem. 1977, 25, 455–459. https://doi.org/10.1021/jf60211a025
[23] J. Scherb et al., Quantitation of S-Methylmethionine in Raw Vegetables and Green Malt by a Stable Isotope Dilution Assay Using LC-MS/MS: Comparison with Dimethyl Sulfide Formation after Heat Treatment, J. Agric. Food Chem. 2009, 57, 9091–9096. https://doi.org/10.1021/jf901765q
[24] C. Dawid, T. Hofmann, Identification of Sensory-Active Phytochemicals in Asparagus (Asparagus officinalis L.), J. Agric. Food Chem. 2012, 60, 11877–11888. https://doi.org/10.1021/jf3040868
[25] K. Kojima, N. Sakurai, IAA Distribution in Etiolated Spears of Asparagus, HortScience 1994, 29, 822. https://doi.org/10.21273/HORTSCI.29.7.822
The article has been published in German as:
- Vom Hochgenuss zum Gestank,
Sabine Streller, Klaus Roth,
Chem. unserer Zeit 2018, 52, 112–119.
https://doi.org/10.1002/ciuz.201800853
and was translated by Caroll Pohl-Ferry.
The Smell of Asparagus Urine – Part 1
What substances cause the characteristic smell of asparagus pee?
The Smell of Asparagus Urine – Part 2
Why does asparagus produce odorous compounds?
The Smell of Asparagus Urine – Part 3
How does asparus change when it is cooked?
The Smell of Asparagus Urine – Part 4
Does everybody’s urine smell after eating asparagus?
See similar articles by Klaus Roth published on ChemistryViews.org




