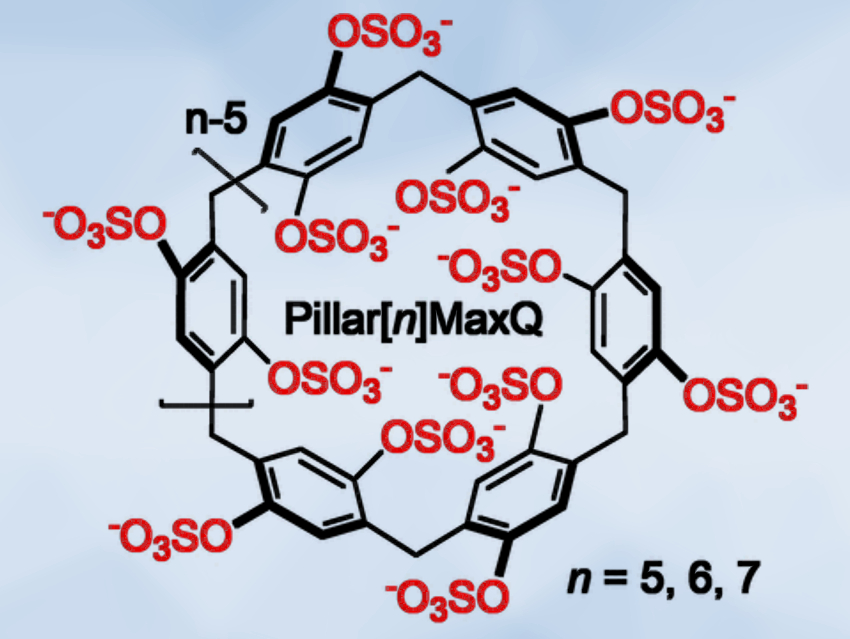
Barrel-Shaped Molecular Containers
Molecular containers that remove drugs, toxins, or malodorous substances from the environment are called sequestering agents. Lyle Isaacs, University of Maryland, College Park, USA, and colleagues have developed a class of molecular containers that specifically sequester neurotransmitter antagonists. The barrel-shaped molecules called Pillar[n]MaxQ (pictured) bind neuromuscular blocking chemicals 100,000-fold more tightly than established macrocyclic detoxification agents.
Molecular containers of the cyclodextrin type sequester their targets by complexation. The ring- or barrel-shaped molecules recognize the molecular features of the target molecules and pull them into the central cavity using hydrophobic forces. Once the target molecule is inside this molecular container, it is neutralized. This host–guest complexation is the mechanism by which cyclodextrins, which are large, ring-shaped sugar molecules, eliminate unpleasant odors.
However, cyclodextrins are not very specific and fail for most alkaloids—a class of nitrogen-containing chemicals, including neurotransmitters and many illicit drugs. For these compounds, a class of molecular containers called pillararenes appear to be more useful. They keep the alkaloids tightly bound in their pillararene cavity by wrapping a ring wall of aromatic benzene units around the hydrocarbon-rich molecular body.
Sequestration of Neurotransmitter Drugs in Water
The researchers have advanced the structure of the pillararenes to make the host–guest interactions stronger and more specific. “We envisioned to create a higher negative charge density around the mouth of the cavity by introducing acidic sulfate functional groups,” they state. The negatively charged sulfate groups attract and bind quaternary ammonium ions, which are a hallmark of several clinically important neuromuscular blocking agents. The sulfate groups also stiffened the molecular structure of the barrels, the team found, so that the drug guest was smoothly pulled into the cavity by hydrophobic forces.
The researchers dubbed the molecular containers Pillar[n]MaxQ, where n indicates a target-size-dependent diameter that is variable. They observed that this class of sequestering agents binds neuromuscular blockers up to 100,000-fold more tightly than the cyclodextrin container Sugammadex, which is in clinical use. Moreover, the sequestering agent discriminated against acetyl choline, a natural transmitter substance of nerve impulses within the central and peripheral nervous systems, which should not be sequestered.
The team measured the host–guest complexation activities of Pillar[n]MaxQ by titration studies involving calorimetry and nuclear magnetic resonance of the guest molecules. As pillararenes have also been shown to reverse the effects of neuromuscular agents in rats, the researchers are aiming to study the new Pillar[n]MaxQ sequestering actions in animal models. Because of the high binding and the specificity of the chemically tailored molecular containers, they are confident that they will observe positive results.
- Pillar[n]MaxQ: A New High Affinity Host Family for Sequestration in Water,
Weijian Xue, Peter Y. Zavalij, Lyle Isaacs,
Angew. Chem. Int. Ed. 2020.
https://doi.org/10.1002/anie.202005902