Atorvastatin, the active ingredient in Lipitor and an effective 3-hydroxy-3-methyl-glutaryl coenzyme A (HMG-CoA)-reductase inhibitor, is in widespread clinical use for the treatment of hypercholesterolemia, high levels of cholesterol in the blood. Given its reliable efficacy for lowering low-density lipoprotein (LDL) cholesterol and its established safety profile, continuous demand is expected and, despite the variety of available strategies for its synthesis, the development of more efficient and scalable syntheses is of continual interest.
Naoya Kumagai, Masakatsu Shibasaki and co-workers, Institute of Microbial Chemistry, Tokyo, Japan, have developed a second-generation synthetic route to atorvastatin that has eliminated five synthetic steps, largely by retention of the amino functionality in a thioamide and implementation of an oxy-Michael reaction to construct and protect the syn-1,3-diol.
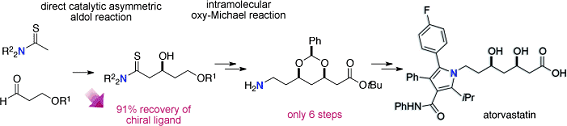
Furthermore, the strategy, which is based on the direct catalytic, asymmetric aldol reaction of a thioamide, allows the efficient recovery (91 %) and reuse of the expensive chiral ligand used for the initial aldol reaction. Further optimization of each step for industrial applications is currently under way.
- Streamlined Catalytic Asymmetric Synthesis of Atorvastatin,
Yuji Kawato, Sandeep Chaudhary, Naoya Kumagai, Masakatsu Shibasaki,
Chem. Eur. J. 2013.
DOI: 10.1002/chem.201204609