Since the advent of frustrated Lewis pairs (FLPs) in 2006, the most commonly studied Lewis acids for this chemistry have been based on B(C6F5)3 or closely related electrophiles, or intramolecular systems in which the B(C6F5)2 fragment is appended to a donor. Such FLPs have been employed in catalytic metal-free hydrogenation and to activate a number of small molecules, including H2, alkenes, CO2, SO2, CO, and NO. Expansion of FLP chemistry to transition-metal species has also attracted attention in recent years. However, in all cases to date, the metal center acts as the Lewis acidic center.
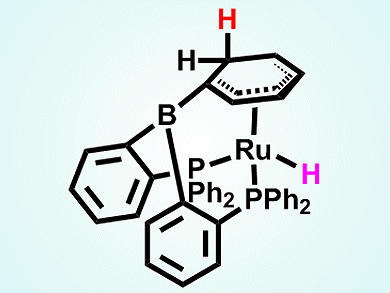
Michael Boone and Douglas Stephan, University of Toronto, Canada, were interested in the potential of a metal center to act as an electron-withdrawing group, generating Lewis acidity at a coordinated ligand. To such ends, they recently made [(Ph2PC6H4)2B(η5-C6H5)RuCl][B(C6F5)4], a 18-electron metal cation capable of behaving as a Lewis acid at an aromatic carbon atom [1].
The team now shows that treatment of [(Ph2PC6H4)2B(η5-C6H5)RuCl][B(C6F5)4] with mono and multidentate phosphorus Lewis bases afforded the Lewis acid–base adducts with the ortho-carbon atom of the coordinated arene ring. Similar reactivity was observed upon treatment with N-heterocyclic carbenes. However, adduct formation occurred at both ortho– and para-carbon atoms of the bound arene with the para position being favored by increased steric demands.
This metal-based Lewis acid can effect the heterolytic activation of H2 with the hydride being added exclusively to the ortho-position of the coordinated arene (pictured).
This is the first reported system in which an aromatic carbon behaves as a Lewis acid in FLP chemistry.
- Ru–η6-Arene Cations [{(Ph2PC6H4)2B–(η6-Ph)}RuX]+ (X=Cl, H) as Lewis Acids,
Michael P. Boone, Douglas W. Stephan,
Chem. Eur. J. 2014.
DOI: 10.1002/chem.201304289
[1] M. P. Boone, D. W. Stephan, J. Am. Chem. Soc. 2013, 135, 8508–8511. DOI: 10.1021/ja403912n