Scientists in China have described the Kumada-Tamao-Corriu reaction of phenolic salts with various aryl Grignard reagents using a nickel catalyst. The fact that phenols, in place of aryl halides, can be used directly in cross-coupling chemistry is a significant finding.
Zhang-Jie Shi and co-workers have used this method to prepared biaryl scaffolds, which are important in material chemistry and the construction of natural products. Extra steps of group transfer and the generation of organic wastes were avoided by a direct transformation from the magnesium phenolate itself. Preliminary studies were conducted to understand the reaction mechanism.
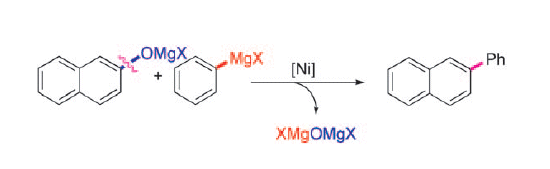
- Direct Application of Phenolic Salts to Nickel-Catalyzed Cross-Coupling Reactions with Aryl Grignard Reagents
D.-G. Yu, B.-J. Li, S.-F. Zheng, B.-T. Guan, B.-Q. Wang, Z.-J. Shi
Angew. Chem. 2010, Early View