Can an image capture the dual behavior of light at the same time?
It is well known that light can behave as a particle and as a wave, but it took until 2015 to capture this dual behavior of light simultaneously in an image [1].
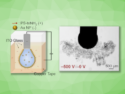
Fabrizio Carbone and his team at the École polytechnique fédérale de Lausanne (EPFL), Switzerland, fired a pulse of infrared laser light at silver nanowires, which were located on a layer of multilayered graphene. The laser light stimulated electronic plasma oscillations in the nanowires, as it adds energy to the charged particles in the nanowire, thus causing them to oscillate. This electromagnetic far field is accompanied by an electromagnetic near field located directly on the wire surface. Light travels back and forth on the nanowire. When waves traveling in opposite directions meet, they form a standing wave.
To image this standing wave of light, the scientists shot a stream of electrons close to the nanowire. As the electrons interacted with the confined light on the nanowire, they either sped up or slowed down. By using an ultrafast microscope, Carbone’s team imaged the position where this change in speed occurred. By doing this they were able to visualize the standing wave, which acts as a fingerprint of the wave-nature of light.
At the same time, this phenomenon shows the particle-like character of light: As the electrons pass close to the standing wave of light, they hit the photons. This affects their speed, and this change in speed appears as an exchange of energy packets, called quanta, between electrons and photons.
The picture showing this dual behavior resulted in quite a bit of media hype not only in scientific circles but also in general publications like the New York Times. Professor Fabrizio Carbone talked with Dr. Vera Koester of ChemViews magazine about the research behind the picture.
Professor Carbone, you captured the image using EPFL’s ultrafast energy-filtered transmission electron microscope. In a nutshell, what is special about this microscope and why did you use it?
The difference between what we do and what is commercially done in microscopy is analogous to the difference between a still camera and a video recorder. However, because we are looking at atoms, the timescale is not measured in milliseconds as with a video recorder but in femtoseconds — each snapshot that we take with our “movie camera” is 200 femtoseconds in duration. This is important for the experiment: Light never stops, so if you want to confine light in time and space, you have to take a snapshot very quickly. That was the reason for doing this experiment with the ultrafast microscope.
Was your study directly aiming to look at the wave–particle duality?
Originally it was not. We do these experiments to characterize the confined electromagnetic fields on nanostructures. Initially, the purpose of this experiment was to study new optoelectronic materials and devices. However, when this effect – which allows us to record images of electromagnetic fields – was discovered around 2008 at Caltech where I then used to work, I actually said to Professor Zewail, my advisor at that time who also received the 1999 Nobel Prize in Chemistry for his work on femtochemistry, that I thought it could be a very good way to see quantization and interference of an electromagnetic field. So the idea has been in the back of my mind for a long time.
What is special about the image?
The picture is not like a commercial photograph, which happens in ordinary space, in x and y coordinates. If you do this with light, you can take very good image of light in an interference pattern or something. But when you want to explore the quantum behavior of light, light quantization, you need to look into a different dimension that is not space but, for example, energy.
After the media picked up the picture, a lot of people mistakenly took these data as a revelation of something that has been debated since the beginning of the century: whether you can really see the two complementary effects of light at the same time or not.
What we have achieved is a nice way of imaging the electromagnetic field but it does not necessarily add anything to what has already been done or is known. The purpose of our experiment was to set up an experimental apparatus to take this image in space and energy. The real breakthrough here is the image itself. However, we have other data that support the discussion about whether you can see both effects at the same time.
Why hasn’t anybody ever done this before?
Because it is not easy at all. First, there are only two microscopes capable of doing such experiments, one in our lab and one at Caltech. Second, we took this image at least 20 times. When we got the first one, we understood that it worked, but it would have been very hard to convince anybody of what we were seeing on it. So then we optimized the system. This basically had to do with reshaping the beam in a way that it matched the shape of the wire as much as possible, which meant focusing the electron beam very tightly, and finding a wire with a very good resonance condition that supports a lot of light. We finally took the picture shown in the press release last July. We reproduced these data many times, but it is very hard to reproduce it as well as it is shown in the picture – everything was perfect there.
Were there similar experiments?
There was a so-called breakthrough experiment in 2011: The simple double-slit experiment showed that as long as light is unobserved, each individual particle seems to go through both slits of the gap. It forms an interference pattern behind the gap. However, this disappears as soon as one tries to gain information about the path of the particle. A consequence of this quantum mechanical uncertainty is that one cannot discuss the path or “trajectory” that a quantum particle takes. In the experiment, single photons emitted by a quantum dot were sent through a double-slit interferometer [2]. Their trajectories were reconstructed by performing a weak measurement of the photon momentum. However, it was pointed out that these trajectories cannot necessarily be related to one single photon. So it wasn’t the final answer to the question.
In our experiment you have a bunch of photons that you put on this wire and you can see interference and quantization. So even if you can interpret this strictly speaking as a multiparticle experiment where you have several photons and some of them behave as particles and some of them as waves, you might wonder why some of them choose one way and the other the other way if the experiment was all the same. So that is really the interesting question.
Why is the image important for you?
When I was at school, they taught me that light is a wave and that you have plenty examples of that — you can even do experiments in the kitchen that show you that. Then they tell you that the photoelectric effect shows that light is also a stream of particles, each of which has this energy package: ħω. This effect is not that great; you don’t see it in the kitchen, and it requires interpretation of a graph and all that to understand it. Then they tell you that the two things happen at the same time. This is great but it is a nightmare to explain. So my thinking was to explain this aspect to people graphically.
How did your students react to the media hype about the picture?
We are a small and very young group. When the paper came out, we all went out for dinner together as a celebration.
I started my group here five years ago, so everybody was hired more or less at the same time. There are currently three students who are graduating, and one of them had this result. To me it is not the best result we have had in the lab, but it is certainly the one that sells the best. I think the merit is also in the way it was conceived: It was a good idea to publish it as a picture of an electromagnetic field. This interests people because it is visual and one has the feeling of being able to grasp what it is about more quickly.
Another student had her thesis nominated for an award for the best thesis, so she has had equally good results. Maybe they will sell less well when they are published — that is, maybe they are less appealing to the general public, but scientifically they are very good results. And she already has a very high-profile job secured in the United States. So I think everybody is happy.
That’s great. Thank you very much for the interview.
[1] Simultaneous Observation of the Quantization and the Interference Pattern of a Plasmonic Near-Field,
L. Piazza, T. T. A. Lummen, E. Quiñonez, Y. Murooka, B. W. Reed, B. Barwick, F. Carbone, Nature Communic. 2015. DOI: 10.1038/ncomms7407
[2] Observing the Average Trajectories of Single Photons in a Two-Slit Interferometer,
S. Kocsis, B. Braverman, S. Ravets, M. J. Stevens, R. P. Mirin, L. K. Shalm, A. M. Steinberg,
Science 2011, 332, 1170–1173. DOI: 10.1126/science.1202218

Fabrizio Carbone gained his PhD in condensed matter physics from the University of Geneva, Switzerland, under the supervision of van der Marel in January 2007. He performed postdoctoral work in physical chemistry at the California Institute of Technology (Caltech), Pasadena, CA, USA, in the group of Ahmed Zewail till 2009. Since 2010, Carbone is Tenure Track Assistant Professor at the École polytechnique fédérale de Lausanne (EPFL), Switzerland.
Selected Publications
- Coupling of a high-energy excitation to superconducting quasiparticles in a cuprate from coherent charge fluctuation spectroscopy,
B. Mansart, J. Lorenzana, A. Mahn, A. Odeh, M. Scarongella,M. Chergui, F. Carbone,
Proc. Natl. Acad. Sci. USA 2013, 110(12), 4539–4544.
DOI: 10.1073/pnas.1218742110 - Principles and Implementation of an Ultrafast Transmission Electron Microscope,
L. Piazza, M. Cottet, D. J. Masiel, T. LaGrange, F. Carbone,
Microsc. Microanal. 2012, 18 (Suppl 2), 600–601.
DOI: 10.1017/S1431927612004850 - A perspective on novel sources of ultrashort electron and X-ray pulses,
F. Carbone, P. Musumeci, O. J. Luiten, C. Hebert,
Chem. Phys. 2012, 392, 1, 1–9.
DOI: 10.1016/j.chemphys.2011.10.010
Also of interest
- International Year of Light 2015 (IYL 2015)
A global initiative adopted by the United Nations to raise awareness of how optical technologies promote sustainable development and provide solutions to worldwide challenges in energy, education, agriculture, communications, and health. - Plasmonen als Wellen und Teilchen (in German),
Rainer Scharf,
ProPhysik 5 May 2015. - Elektronen steuern mit Licht (in German),
Ralf Vogelgesang, Jan Vogelsang, Christoph Lienau,
Physik J. 2015, 6, 20.