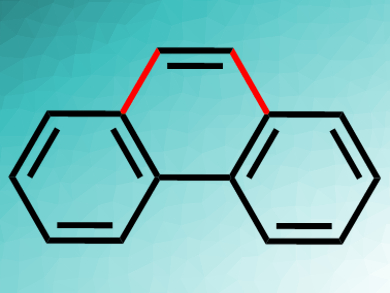
Phenanthrene (pictured) is one of the simplest polycyclic aromatic hydrocarbons (PAHs) and a useful building block for a range of materials. It can be synthesized from 2-biaryl Grignard reagents and alkynes in an iron-catalyzed reaction. However, this reaction needs two equivalents of Grignard reagent, since one equivalent is “sacrificed” as a hydrogen acceptor.
Jianming Yan and Naohiko Yoshikai, Nanyang Technological University, Singapore, have developed a chromium-catalyzed reaction protocol for the synthesis of phenanthrenes from 2-biaryl Grignard reagents and alkynes that uses excess alkyne as the hydrogen acceptor. The team prepared a 2-biphenylmagnesium bromide−lithium chloride complex from 2-bromobiphenyl and Mg·LiCl. Then, they used CrCl2 as a catalyst, 2,2′-bipyridine (bpy) as a ligand, and tetrahydrofuran (THF) as the solvent and combined this Grignard species with a range of alkynes to give the corresponding phenanthrenes.
The reaction tolerates a variety of substituents at the alkyne and at the biaryl species and proceeds in moderate to good yields. Based on the reaction’s selectivity and deuterium labeling experiments, the researchers propose a mechanism that involves a 2-biarylchromium species, which is converted to a metallafluorene hydride. The alkyne is then inserted into the Cr−H bond. Excess alkyne acts as a hydrogen acceptor, which, according to the team, makes the reaction mechanistically unique.
- Phenanthrene Synthesis via Chromium-Catalyzed Annulation of 2-Biaryl Grignard Reagents and Alkynes,
Jianming Yan, Naohiko Yoshikai,
Org. Lett. 2017.
DOI: 10.1021/acs.orglett.7b03342