Cisplatin, one of the earliest and most studied chemotherapeutic drugs, has long been shown to cause DNA crosslinks. Although this valuable cancer drug has saved many lives, treatment with cisplatin comes with a range of serious side effects. Because it is administered generally, the side effects affect the whole body, from kidney damage to nausea to hearing loss. Thus, researchers have worked to develop less harmful platinum complexes that can be targeted to the affected area. Currently, the third generation of platinum drugs is being developed. These drugs are less toxic and photoactivated so that therapy can be applied at a specific position using UV light.
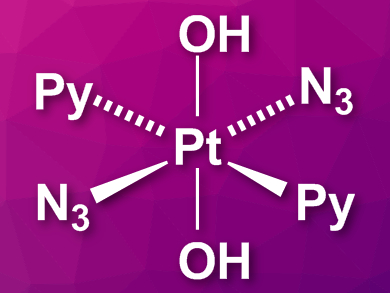
Peter J. Sadler, Peter B. O’Connor, and colleagues, University of Warwick, UK, have developed an octahedral platinum(IV) complex, trans,trans,trans-[Pt(N3)2(OH)2(pyridine)2] (pictured), which has already been shown to inhibit tumor growth upon activation with blue light. Initial studies of the complex’s mechanism of action showed reactivity with DNA and generation of radicals. The team has studied the effects of the complex on peptides, which serve as models for the interaction with proteins in cells.
Using state-of-the-art mass spectrometry techniques, they tested the reactivity of the complex with two model peptides. The results are striking: The complex reacts with various amino acid side chains upon photoactivation. In particular, it seems to target histidine, lysine, and arginine residues, but the frequency of each of these was sequence-dependent. In addition, the team found that methionine and tryptophan residues were oxidated via a radical mechanism, which could have important implications for cancer cells that often exist in a hypoxic environment. Overall, the results show that the complex could be effective at inhibiting cancer, not only due to the formation of DNA crosslinks but also by reacting with proteins and causing oxidative damage.
- Sequence-dependent attack on peptides by photoactivated platinum anticancer complexes,
Christopher A. Wootton, Carlos Sanchez-Cano, Andrea F. Lopez-Clavijo, Evyenia Shaili, Mark P. Barrow, Peter J. Sadler, Peter B. O’Connor,
Chem. Sci. 2018.
https://doi.org/10.1039/c7sc05135b