Carbon nanotubes (CNTs) are well‐known nanomaterials. They have been extensively studied and used in various applications. Nitric acid is often used to treat CNTs in purification processes or to prepare oxidized CNTs. However, little attention has been paid to the effect of HNO3 treatments on the optical properties of CNTs.
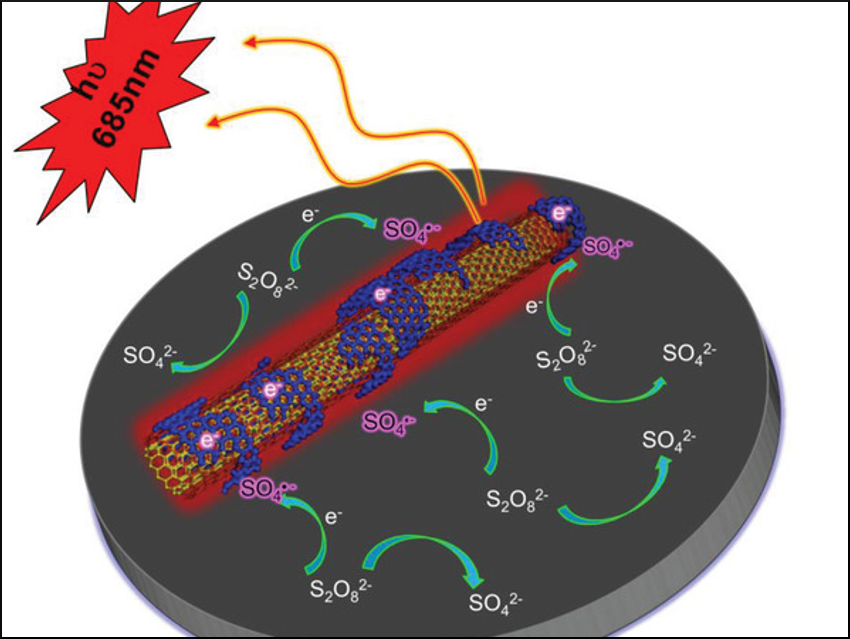
Yuwu Chi and colleagues, Fuzhou University, China, have observed for the first time that HNO3‐oxidized multiwalled carbon nanotubes (ox‐MWCNTs) have strong electrochemiluminescence (ECL) activity. The team treated raw MWCNTs with HNO3 and suspended them on a glassy-carbon electrode. They found that ox‐MWCNTs showed a strong ECL effect with a broad emission spectrum and long maximum emission wavelength (685 nm) in the presence of an electrolyte containing K2S2O8. The ECL emission activity is highly dependent on the oxidation degree of MWCNTs: the higher the oxidation degree, the stronger the ECL emission.
The team used characterization technologies such as transmission electron microscopy (TEM), X‐ray photoelectron spectroscopy (XPS), and Raman spectroscopy to investigate the cause of the ECL activity. According to the researchers, the ECL emission results from immobilized graphene quantum dots (GQDs) on the nanotubes’ surface (pictured in dark blue). The nanotubes could have applications in ECL sensing and in the study of CNT‐based catalysts.
- Strong Electrochemiluminescence Emission from Oxidized Multiwalled Carbon Nanotubes,
Ruina Wang, Haishan Wu, Rui Chen, Yuwu Chi,
Small 2019.
https://doi.org/10.1002/smll.201901550