Rigid organic structures can be useful, e.g., to improve the properties of pharmaceutically active compounds. [1.1.1]Propellane is one example of such a rigid structure. It can be used as a linker to give bicyclo[1.1.1]pentanes. This compound can, e.g., be inserted into disulfide bonds. However, examples of this type of reaction are rare.
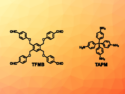
Stefan Bräse, Karlsruhe Institute of Technology (KIT), Germany, and colleagues have investigated this type of reaction and developed a protocol for the synthesis of 1,3-bissulfanylbicyclo[1.1.1]pentanes (pictured) from disulfides and [1.1.1]propellane. The team used UV light as a radical initiator and reacted a variety of aromatic disulfides and several aliphatic disulfides with [1.1.1]propellane at room temperature.
Depending on the concentration and mixture of disulfides, the team obtained either symmetrically or unsymmetrically substituted 1,3-bissulfanylbicyclo[1.1.1]pentanes, as well as the corresponding [2]staffanes (molecules with a central chain of two bicyclo[1.1.1]pentanes) as a side product. The selectivity of the reaction can be tuned by modifying the conditions.
- Insertion of [1.1.1]propellane into aromatic disulfides,
Robin M. Bär, Gregor Heinrich, Martin Nieger, Olaf Fuhr, Stefan Bräse,
Beilstein J. Org. Chem. 2019, 15, 1172–1180.
https://doi.org/10.3762/bjoc.15.114