The current worldwide outbreak of the respiratory disease COVID-19 is caused by the coronavirus SARS-CoV-2. To multiply, the virus has to copy its genetic material inside a host cell. This process is performed by an enzyme, the virus’s RNA-dependent RNA polymerase (RdRp). This polymerase binds to the virus RNA and copies it. Drugs that disrupt this replication process could be useful to treat COVID-19. One such drug, remdesivir, is currently being studied clinically. Knowing the structure of the polymerase of SARS-CoV-2 during replication could provide useful insights for optimizing this type of drug.
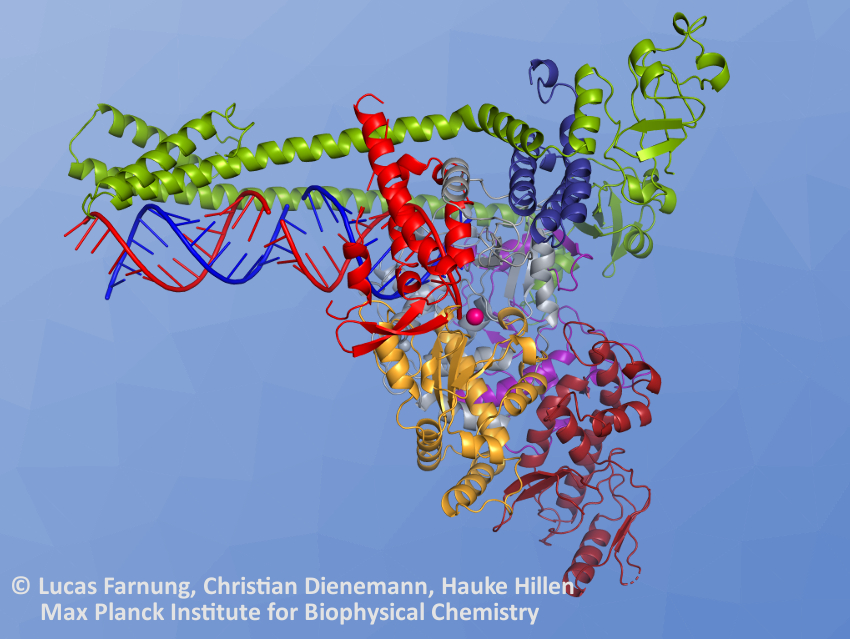
Patrick Cramer and colleagues, Max Planck Institute for Biophysical Chemistry, Göttingen, Germany, have determined the structure of the SARS-CoV-2 RdRp in its replicating form (pictured) using cryogenic electron microscopy (cryo-EM). Cryo-EM uses very low temperatures to allow the determination of biomolecular structures at almost atomic resolution. The polymerase consists of three viral proteins—called nsp12, nsp8, and nsp7. The team expressed these proteins in insect cells or Escherichia coli bacteria, purified them, and reconstituted the polymerase from the purified proteins. Then they added model RNA to form an RdRP–RNA complex and performed the cryo-EM measurements.
The RNA template and the copied RNA product form a duplex, analogous to the double helix of DNA (pictured in blue and red on the left side of the structure). The structure the researchers determined contains over two “turns” of this RNA duplex, in addition to the enzyme’s proteins. Long helical extensions in the nsp8 protein (pictured in green above the RNA) protrude along the exiting RNA duplex. According to the researchers, these protein “extensions” might help the enzyme to copy the relatively long genome of SARS-CoV-2 in a continuous process by preventing the dissociation of the product.
- Structure of replicating SARS-CoV-2 polymerase,
Hauke S. Hillen, Goran Kokic, Lucas Farnung, Christian Dienemann, Dimitry Tegunov, Patrick Cramer,
bioRxiv 2020.
https://doi.org/10.1101/2020.04.27.063180The research has been published as a preprint and has not yet been peer-reviewed. It should not be used to guide clinical practice.
Also of Interest
- Collection: SARS-CoV-2 Virus
What we know about the new coronavirus and COVID-19 - LitCovid
Curated literature hub for tracking up-to-date scientific information about COVID-19 - Many publishers and other entities have signed a joint statement to ensure that COVID-19 research findings and data are shared rapidly and openly