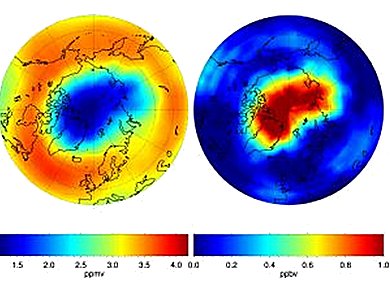
Björn-Martin Sinnhuber, KIT Institute of Meteorology and Climate Research (IMK), Karlsuhe, Germany, and colleagues detected that ozone degradation above the Arctic for the first time reached an extent comparable to that of the ozone hole above the South Pole about a year ago. Occurrence of the Arctic ozone hole was mainly due to the extraordinarily cold temperatures in the ozone layer that is located at about 18 km height in the stratosphere, i.e., the second layer of the earth’s atmosphere. There, chlorine compounds originating from chlorofluorocarbons (CFC, e.g., greenhouse gases and refrigerants) and other pollutants are converted at temperatures below –78 °C to conversion products that attack the ozone layer and destroy it partly. One of the main statements in the studyis that if the trend to colder temperatures in the stratosphere observed in the past decades will continue, repeated occurrence of an Arctic ozone hole has to be expected.
Further decrease in temperature by just 1 °C would be sufficient to cause a nearly complete destruction of the Arctic ozone layer in certain areas. Further development of the ozone layer will consequently be influenced also by climate change. The increase in carbon dioxide and other greenhouse gases will warm up the bottom air layers near the ground due to the reflection of part of the thermal radiation by the bottom layer of the atmosphere towards the earth’s surface, but also result in a cooling of the air layers of the stratosphere above, where the ozone layer is located.
Image: © IMK-ASF, KIT
- Geophysical Research Letters 2012, 38.
DOI: 10.1029/2011GL049784