The olefin (alkenyl) group is perhaps the most useful in synthetic organic chemistry. Moreover, nature finds it easy to enzymically desaturate the relatively inert aliphatic unit (alkyl). The removal of hydrogen from an aliphatic carbon chain to produce an isolated olefin (desaturation) without overoxidation is one of the most impressive and powerful biosynthetic transformations. There are no simple and mild laboratory substitutes.
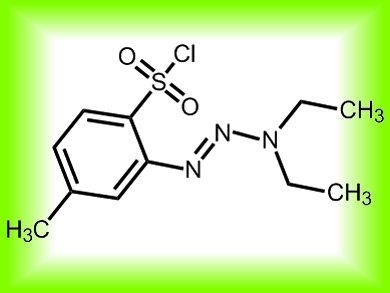
A team at the Scripps Institute, California, USA, have developed what they refer to as a “portable desaturase” (pictured). The artificial enzyme, from o-tosyl triazene chloride, works under mild conditions and is metal free, so is amenable to sensitive reaction schemes. It converts simple derivatives of saturated alcohols and amines into their olefinic counterparts in a predictable way without causing troublesome overoxidation. Despite its simplicity it can be used in complex reaction schemes without interfering with other functional groups.
- Guided desaturation of unactivated aliphatics,
Ana-Florina Voica, Abraham Mendoza, Will R. Gutekunst, Jorge Otero Fraga, Phil S. Baran,
Nature Chem. 2012.
DOI: 10.1038/nchem.1385