Everywhere you can read about the shale gas rush in North Dakota, Texas, and Montana, all USA. USA is the largest producer of shale gas. Their shale gas production rose from 2000 to 2010 from 2 to 23 % overall gas production with the trend still rising [1].
How does hydraulic fracturing (fracking) and horizontal drilling make natural gas trapped in shale rock formations financially feasible and accessible?
Shale Gas
Shale is a compressed fine-grained type of sedimentary rock. It was formed from mud silt, clay, and organic matter.
Shale gas is natural gas trapped within tiny pore spaces in shale formations. It is a hydrocarbon gas mixture. It consists mainly of methane. Other hydrocarbons are natural gas liquids (NGLs) like ethane, propane, and butane, and it also contains carbon dioxide, nitrogen, and hydrogen sulfide.
Horizontal Drilling and Fracking
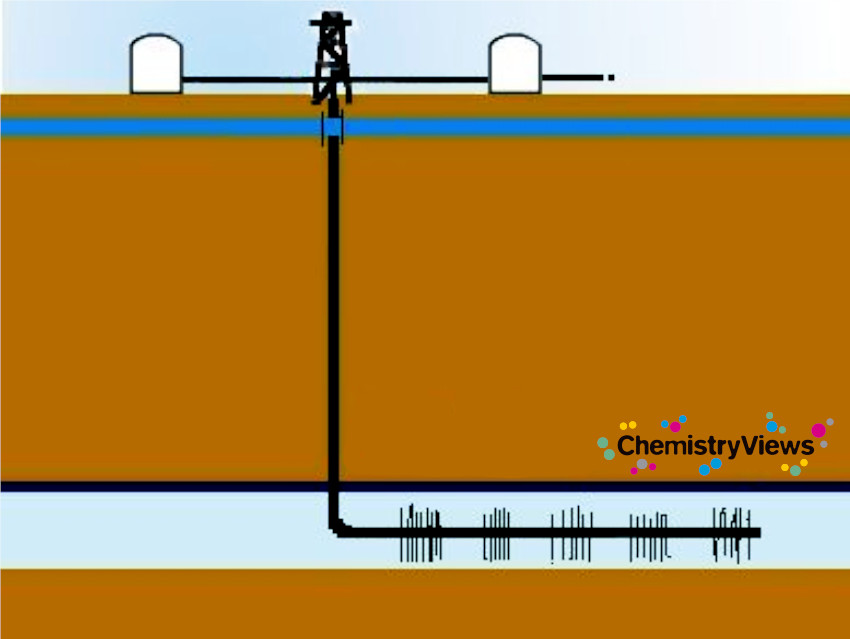
Horizontal drilling allows drilling vertically several thousand meters (typically 6,000 m) deep and then turning 90 ° and drilling horizontally. This makes it possible to operate multiple target zones from one drilling pad. It enlarges recoverable reserves and also productivity is significantly increased.
Hydraulic fracturing or fracking creates fractures in the shale formation to release the gas.
A fracturing fluid is pumped under high pressure (ca. 100 bar) into the drilling pipe to widen fractures in the rock or to create new ones. The fluid consists mainly of water. It is usually mixed with quartz sand and three to twelve chemicals which amount for 0.5 to 2 %. The exact compositions are kept secret by the drilling companies.
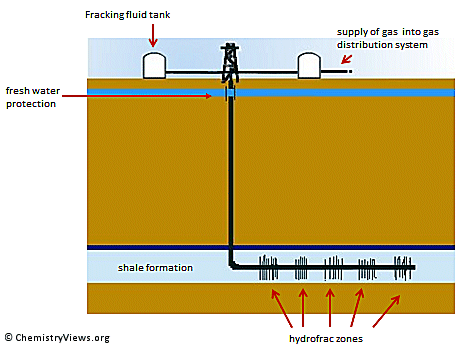
Approximately 7–15 million L of water are used per well [2]. The sand holds the cracks open to increase the amount of natural gas that can be extracted. The chemicals may include gels or butyldiglycol (typical 0.2 L/t water) that increase the viscosity of the fracking fluid to better transport the sand, foaming agents like CO2 and N2, to transport the sand, biocides to prevent growth of bacteria at organic components, acids like HCl, acetic acid, formic acid or boric acid to disintegrate the minerals of the rock formations, and anti-corrosion agents to protect the site when acid is added.
Approximately 50–70 % of the water solution is recovered. The remaining water stays in the ground. The recovered water is contaminated with chemicals and salts from the rock formation. It is either transported or directly disposed in treatment facilities.
What to do With Shale Gas?
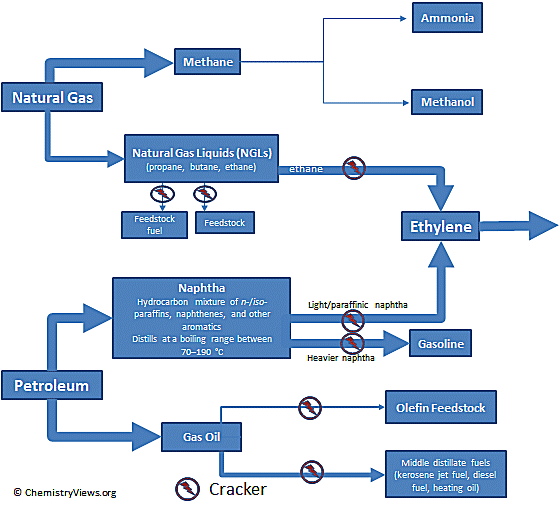
Cracking means to separate the large hydrocarbon chains of fossil fuels like natural gas and petroleum. Naphtha is related to the price of oil, which is set by global supply and demand. The natural gas prices are regional. Ethylene is the most widely used organic compound in chemical industry.
In the USA, over 85 % of ethylene is derived from natural gas. In Western Europe, over 70 % is derived from naphtha and other light distillated oil-based products. Therefore, the shift toward more and lower-cost natural gas has benefitted the chemical industry in the USA. Here, it currently costs $ 300 to produce 1 ton of ethylene. In Asia it costs $ 1,717 and in Saudi Arabia $ 455 [3, 4].
Concerns
Among the environmental concerns are that enormous amounts of water are used and the water is contaminated by the layers of earth it is pumped through as well as the fracking chemicals.
The cement to protect the ground water can get leaky with time, especially if acid water is used.
Radon and mercury are gases which can leak out from the shale formation. Other subsurface materials like lead or arsenic may be mobilized.
It cannot be excluded that fractures in rock formations grow in higher rock/soil formations or up to the surface.
Small earthquakes, so called induced seismicity, might result from changing the balance of forces in the rock formations.
There also are the concerns regarding energy politics, fearing that the hype can change the understanding that shale gas is a transition and not a replacement fuel.
Also methane, which is the main component of natural gas, is a potent greenhouse gas. It is poorly understood what impact increasing shale gas industry has on the climate. [1, 2]
References
[1] A. Kotsakis, The Regulation of the Technical, Environmental and Health Aspects of Current Exploratory Shale Gas Extraction in the United Kingdom. Initial Lessions for the Future of European Union Energy Policy, Rew. Europ. Com. Int. Environm. Law 2012, 21 (3), 282–290. DOI: 10.1111/j.1467-9388.2012.00759.x
[2] L. Reins, The Shale Gas Extraction Process and Its Impacts on Water Resources, Rew. Europ. Com. Int. Environm. Law 2011, 20 (3), 300–312. DOI: 10.1111/j.1467-9388.2012.00733.x
[3] Kevin Bukkis, Shale Gas Will Fuel a U.S. Manufacturing Boom, MIT Technology Review, January 9, 2013.
[4] Shale Gas and New Petrochemicals Investment: Benefits for the Economy, Jobs and US Manufacturing, American Chemistry Council (ACC), March 2011.
Also of Interest
- Shale Gas: Impact on the Petrochemical Industry,
ChemViews magazine 2013.
DOI: 10.1002/chemv.201300015
Development of shale oil and gas has opened new sources of oil, natural gas, and key petrochemical feedstocks - Shale Oil Worldwide,
ChemistryViews.org 10 December 2012.
US and Canada seem to remain the main countries to take economic advantage of shale development for some time