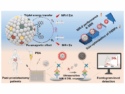
Quantum dots and rare-earth-metal-based nanoparticles are two of the most widespread fluorescence systems developed for sensing and imaging applications. Carbon-based materials like carbon nanotubes, graphene dots, and carbogenic nanodots (CNDs) have emerged as alternatives owing to their less toxic nature and simpler synthetic routes. CNDs are particularly interesting because some CNDs are able to convert longer wavelength radiation (in the near IR range) to shorter wavelengths (in the visible range). This property known as “up-converting” maks them ideal for applications such as bioimaging, sensing, photocatalysis, and in solar cells.
Jose L. Hueso, University of Zaragoza, Spain, and Networking Research Center on Bioengineering, Madrid, Spain, and colleagues fabricated CNDs with interesting properties from vitamin C. They used hydrothermal carbonization in the presence of ethanolamine as a nitrogen source to make N-doped CNDs (N-CNDs).
The team tested the photophysical properties of the N-CNDs and compared them to CNDs synthesized without ethanolamine. N-CNDs showed the ability for up-conversion and a pH-dependent fluorescence lifetime response. The N-CNDs were able to detect copper ions in solution, which quench the intensity of the N-CNDs. Other ions do not affect the intensity of N-CNDs, except for iron in high concentrations.
- Nitrogen-Induced Transformation of Vitamin C into Multifunctional Up-converting Carbon Nanodots in the Visible-NIR Range,
M. Carmen Ortega-Liebana, M. Mar Encabo-Berzosa, M. Jose Ruedas-Rama, Jose L. Hueso,
Chem. Eur. J. 2016.
DOI: 10.1002/chem.201604216