All plants produce considerable amounts of polyphenols as secondary metabolites, which then serve as flower pigments, protection from UV light, and defense against the plant’s being eaten. Polyphenols are regarded as healthy substances since they are thought to shield people from “illnesses of civilization” such as cardiovascular disease, cancer, or diabetes. But what specific compounds show a prophylactic effect with respect to these conditions, and how do they work?
8. How Do We Know That Polyphenols Are Healthy?
First indications in this respect came historically from epidemiological studies [6]. Here, statisticians collect the frequency of appearance of certain diseases among large human populations. If one observes a reduced prevalence of, e.g., cardiovascular disease, cancer, or diabetes in some population, they reason that there might be one of three primary causes:
- The populations differ genetically—e.g., residents of the Indian subcontinent suffer more frequently from cardiovascular problems than Europeans or Asians, and they appear less able than Europeans to tolerate milk products and alcohol.
- A given population may suffer more under adverse environmental influences, e.g., serious air or water pollution.
- It could be that diet is responsible. A suspected reduction in specific diseases might be due to dietary habits.
Cause 3 could be established through epidemiological studies, in which the frequency of a particular condition is shown to correlate with consumption of certain foodstuffs. Through such a study, epidemiologists ascertain the statistical probability of a connection between consumption of a specific food and a decrease or increase in the occurrence of some medical condition.
However, epidemiological studies cannot address a causal relationship, only give a statistical probability that there is a relationship between the condition in question and diet. Obviously, one cannot obtain any information in this way regarding the chemical compound(s) responsible, nor find a mechanism behind the effects.
Epidemiological Studies
The earliest known epidemiological studies on polyphenols were carried out in 1934. Desier Cordier, founder of a famous wine shop, noted that more centenarians (persons over 100 years old) lived in the province of Bordeaux than anywhere else in France, and he organized a “long-life festival” under the motto “Wine, the elixir of life.” (Even today there are more centenarians in France overall than in any other European country; for comparison, for every 10,000 people, there are 102 centenarians in France, but only 45 in Germany).
The two most famous epidemiological studies are the “Zutphen Elderly Study” and the “French Paradox Study”. In the Zutphen study from the 1990s (in the Dutch city of Zutphen), a statistical relationship between a diet rich in fruits and vegetables and a reduced incidence of both cardiovascular disease and cancer was shown for the first time. In the French Paradox Study, it was demonstrated that within Europe, the French unexpectedly suffer one of the lowest rates of cardiovascular disease, despite a diet relatively rich in fats. Their low rate of cardiovascular problems, and thereby their increased life expectancy, was attributed to a high consumption of red wines, which are especially rich in polyphenols, and above all to their major component, resveratrol (20).
Clinical Intervention Studies
After work by epidemiologists, two additional steps are required to establish a relationship between diet and health. In general, as a next step, clinical intervention studies are conducted. In this case, certain human subjects are designated (the use of animal models is also possible, although these are less informative) and fed selective foods or food components, after which a set of measurable, relevant physiological parameters is determined. For example, it could be shown that regular consumption of green tea or chocolate leads to a reduction in blood pressure, an increase in the rate of blood flow, and a reduction in infection indicators in the blood. Similarly, consumption of coffee could be related to a lowering of blood sugar levels [6].
Active Agents and Mechanisms
In a final scientific step, the chemical compounds in foods responsible for the observed health effects must be identified and isolated, and their mechanisms of action clarified. This becomes a joint assignment for chemists, biochemists, and pharmacologists.
9. Discovery of Biologically Active Substances
How does a chemist identify polyphenols relevant to human health? In a first step, all relevant compounds from the plant in question must be separated, and their chemical structures established. With the purified substances, it becomes possible to conduct in vitro enzyme assays and other biological tests that might provide clues about the importance of a compound and its possible mechanism of action.
The approach is analogous to that employed in active substance development in the pharmaceutical industry. There is one important difference, however: a pharmaceutical investigator usually knows at least the biological target molecule he is hoping to inhibit with a new active agent. This might, for example, be an enzyme or a receptor that has something to do with a particular disease, and was for this reason targeted specifically at the outset of the research. This is not the case for a foods researcher, however, whose job is more like looking for a needle in a haystack.
Nevertheless, it has become possible in the last two decades to identify a series of biologically active polyphenols that can inhibit specific enzymes in the human body. For example, it was shown that quercetin (17) inhibits phosphoinositide-3-kinase and toposisomerase II, EGCG (epigallocatechin gallate) (15) inhibits DNA-methyltransferases 1 and 3a, and chlorogenic acids (10) inhibit several enzymes in sugar metabolism.
10. Polyphenols in Food
All plant produce large amounts of polyphenols. Daily human consumption of these compounds is estimated to be roughly 10 g [1, 6].
- What foods are good sources of polyphenols?
- Which structures predominate?
- How are the amounts determined?
Two basic methods are employed: on one hand, a group determination for all phenolic components can be carried out. For this purpose, one employs the Folin-Ciolcalteu reagent, a mixture of phosphomolybdate and tungstate, which turns blue in the presence of phenols and thus permits establishing an overall phenol content. Alternatively, the different phenols in a plant can be separated chromatographically and subsequently quantified individually by UV spectroscopy or mass spectrometry (MS).
Pomegranate is the fruit with the highest absolute phenol content. In the Folin-Ciolcalteu test, it shows the highest value so far recorded for total phenol content, followed by results from green tea and coffee. Other fruits with a high phenol content are apples, onions, berries, artichokes, tomatoes, almonds, and apricots. High phenol content is indicated by the tendency of a plant toward browning. This brown coloration is due to an oxidation reaction of phenols, which takes place, for example, after the destruction of plant tissue, as by cutting an apple.
If one considers the overall amount of phenols that an average European consumes in a day, one arrives at the following rather surprising picture for the various foodstuff sources (see Fig. 5):
coffee > fruit juice > tea (green and black) > chocolate > red wine ≥ fruits ≥ vegetables ≥ cereals ≥ other.
The reader interested in detailed data is referred in this context to the “Phenol Explorer” database, which provides essentially all currently available quantitative data on polyphenols in foods [7].
.jpg) |
|
Figure 5. Phenolic substances in selected foodstuffs. (Compound structures see Fig. 2) |
Bioavailability
A food component can behave in the body just like a medicinal product. In order to deploy its effect, any such compound must first enter the bloodstream and then be transported to a site of action (a specific organ or tissue). If this occurs, one regards that compound as “bioavailable”.
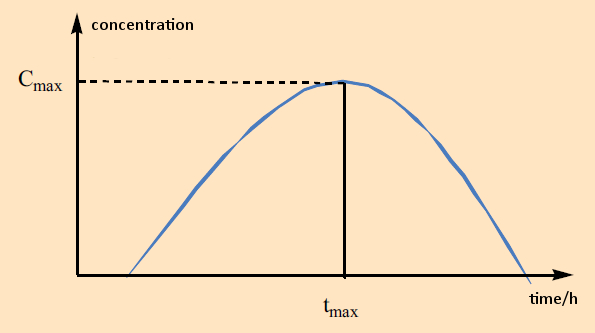
Although processes by which certain polyphenols act on human enzymes had long been known, studies of bioavailability have begun only in the last ten years. First results were quite surprisin because, on the basis of these studies, only very few polyphenols derived from food sources actually become significantly bioavailable. This phenomenon is described by a “Cmax parameter”, characterizing the maximal plasma concentration of a compound in the bloodstream after its consumption. From the time tmax at which a value Cmax is achieved, one can draw certain conclusions about the site within the digestive tract where the absorption of the compound occurs (e.g., 5–10 min: in the mouth; up to 30 min: in the stomach; 30 min–2 h: in the small intestine; 2–10 h: in the colon) (see Fig. 6).
 |
|
Figure 6. Bioavailability of polyphenols (substance concentrations measured in human plasma). |
The highest Cmax values are in the range of 1–2 μM for the isoflavones genistein and daidzein from soy. However, most polyphenol results fall in the nanomolar range, and many—like those for the anthocyanides from berries or the proanthocyanidines from red wine—reveal almost no bioavailability whatsoever. For such substances, it is thus utterly irrelevant whether they perhaps reveal stunning effectiveness in an enzyme test, because they will never make their way into the human bloodstream [6]! Nevertheless, it is indisputable that foods containing these polyphenols do still have an impact.
Antioxidants and Radical Scavengers
Due to their chemical structures, all polyphenols can be regarded as electron-rich. Thus, they are easily oxidized, and can act as antioxidants. Oxidation transforms them into stable free radicals, making them capable of quenching harmful biological radicals (see Fig. 1).
Radicals like the hydroxyl radical •OH and the peroxide radical •OOH arise in all cells through cellular respiration—increasingly so with smokers—and can cause damage to those cells, for example, via DNA mutation or destruction of proteins and lipids, leading in turn to disease. Antioxidants were long thought to unfold their positive effects by capturing free radicals as these are formed (“antioxidant hypothesis”).
In recent decades, the antioxidant hypothesis has been commonly used in explaining positive effects of polyphenols on human health. It has also been successfully employed by the foods industry and by manufacturers of nutritional supplements as a marketing strategy for polyphenol-rich foods and food products. Unfortunately, at least in my opinion, the hypothesis is fallacious. How so?
Fallacious Hypothesis
Polyphenols show extremely low bioavailability. It is generally the case that the more polar a polyphenol, the more effective it is as an antioxidant. But this is not consistent with its observed bioavailability. Here, the relationship is in fact exactly the reverse: water-soluble (polar) compounds can pass the lipophilic walls of intestinal cells only with difficulty, and rarely find their way into the bloodstream. Thus, they cannot function there as antioxidants.
If one compares the concentrations of polyphenols in the blood with those of natural antioxidants arising in the human body, such as vitamin C (Cmax = 50 μM), or vitamin E (Cmax = 30 μM), all the phenols taken together never even approach such values. Moreover, most polyphenols present are reduced by bacterial intestinal flora, their potential effectiveness being thereby further diminished.
Polyphenols also show a so-called pro-oxidative effect: as reducing agents, phenols are able to reduce Fe3+ to Fe2+, or Cu2+ to Cu+. The reduced metal ions, in turn, can produce •OH radicals via the Fenton reaction. This mechanism of action is used, for example, in explaining the anticarcinogenic activity of EGCG (15 in Fig. 2). Phenols from green tea produce radicals that are known to kill off cancer cells. Recent studies have shown, however, that the redox status of the human body is unchanged by a polyphenol-rich diet. In particular, the level of isoprostane, a marker compound formed by radicals subsequent to oxidative stress, is unchanged in human urine after ingesting a diet rich in polyphenols.
Alternative Explanations
One can thus safely shelve the antioxidant hypothesis and is forced to seek an alternative explanation for the health effects of polyphenols. New approaches that represent a paradigm shift in polyphenol science and still contribute to an explanation of their biological effects, currently include, for example, the activation of the NF-kB signal pathway, the stimulation of activator protein-1 (AP-1), the activation of phase-II enzymes and of Nrf2, as well as influencing the signal cascade of mitogen-activated protein kinase (MAPK) [6]. Expressed in simplified form, polyphenols irritate and train the human body, in that they engage a series of signal cascades which the body uses for defensive purposes.
Intestinal Microflora, Metabolism, and Food Processing
How can one explain the positive effects on human health of most phenols if they never even reach the bloodstream?
In explaining their effect on human health, there is a need for a radical rethinking. After years of research, it is necessary to admit that most previous work was headed in the wrong direction; new ideas are needed. As everywhere in experimental research, hypotheses are developed, but they may be falsified later, as with the polyphenol research described here.
To explain the positive health effects of polyphenols, there are presently three scientific explanatory approaches: The first two take into account intestinal microflora. Our intestines are inhabited by ca. 1013 bacteria. Polyphenols are ingested by the intestinal bacteria and then intensively metabolized, i.e., chemically transformed. For many intestinal bacteria, polyphenols are toxic, so that they drastically alter the spectrum of intestinal flora present, which might already lead to health-enhancing effects. Furthermore, intestinal bacteria create many new chemical compounds starting from the polyphenols, at least some of which will have health consequences of their own, especially since they are likely to offer greater bioavailability than their precursor substances of plant origin.
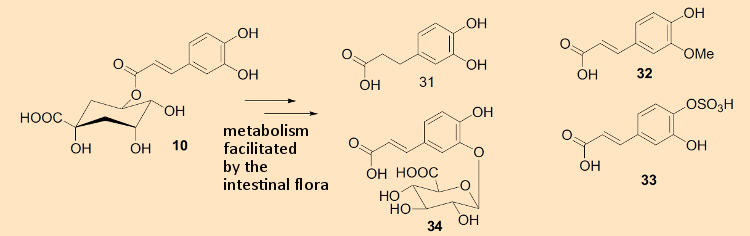
This can be illustrated on the basis of two examples: studies by Alan Crozier have shown that at least ten different metabolites arise by this route from chlorogenic acids in coffee (31–34 in Fig. 2), which are in turn incorporated into the blood (see Fig. 7) [8]. These molecules are further metabolized by various human cells, especially in the liver, leading to the production of yet more new compounds. All these metabolites continue to circulate in the bloodstream and may trigger their own biological reactions.
 |
|
Figure 7. Intestinal flora metabolite of 5-caffeeoylquinic acid, detected in blood plasma from coffee drinkers. |
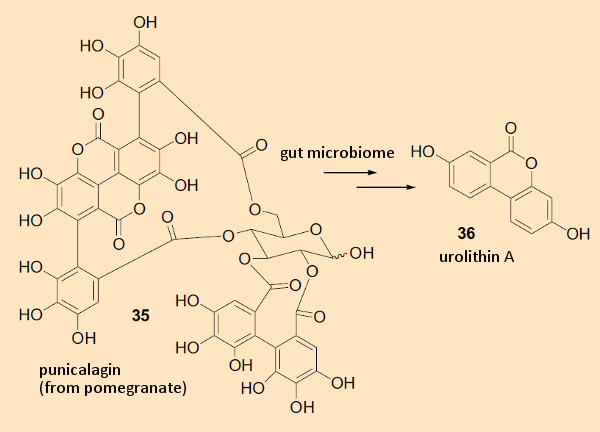
One class of such metabolites has already been shown to have a positive health effect. These are the urolithins (36), formed by intestinal bacteria from pomegranate ellagitannin polyphenols and punicalagin (35) (see Fig. 8) [9]. The compounds are associated with tumor-suppressing activity. This is the reason pomegranate extract is currently being considered for approval as an herbal medication in the treatment of prostate cancer.
 |
|
Figure 8. Intestinal flora metabolite of punicalagin, derived from pomegranates. |
Finally, it is worth considering that humans mostly consume vegetable foodstuffs not in their raw form, but instead after first subjecting them to dramatic chemical changes through some sort of processing, such as boiling, roasting, baking, steaming, toasting, fermenting, pickling, or marinating. The sorts of chemical changes caused by this are in most cases unknown. In our own research, we have shown that starting with only six phenols from a green tea leaf, fermentation leads to 30,000 different polyphenols in a cup of black tea [10], and from 80 phenols in a green coffee bean, about 1,000 new compounds occur as a result of roasting. Biological activity has already been demonstrated for one particular category of these roasting products: the chlorogenic acid lactones. These not only lend a bitter note, but are also capable of binding to human opioid receptors, and thus functioning as painkillers.
We must wait to find out to what extent products of processing, or metabolites, are responsible for further mechanisms of action with respect to polyphenols, but research in this area has already become very exciting.
11. Polyphenol-Containing Medicinal Plants
Polyphenols are present not only in vegetable species, but, of course, also in medicinal plants. Many plants contain high concentrations of materials belonging to this class of compound; indeed, their pharmacological activity can often be traced in large measure to polyphenolic content. Due to their high levels of flavonoids, medicinal plants are in many cases referred to as “flavonoid drugs”. Arnica, birch leaves, cistus, gingko, goldenrod, elderberries, hops, St. John’s wort, camomile, catsfoot (cudweed), lady’s thistle, passionflower, Pelargonium sidoides, marigold, English camomile, pansies, licorice, and hawthorn are among the more familiar—indeed, classical—flavonoid drugs [11, 12].
Other medicinal plants containing large amounts of phenols are especially those from the Lamaceae, Rutaceae, Asteraceae and Rubiaceae families. All these medicinal plants provide general medicinal effectiveness quite similar to that of polyphenol-containing foods. Their activity can be generally characterized as antiallergenic, antiphlogistic, antimicrobial, antiviral, anticarcinogenic, and antioxidative. Consistent with their relatively broad spectrum of activity, these plants are employed in phytotherapy for a wide array of disorders.
Interestingly, a few medicinal plants have been found to display unexpected effects: St. John’s wort is said to be an antidepressive, hops a sedative, pelargonium root extract an immune stimulant, and ginkgo an antidementive. These particular plants also have in common that a definitive mechanism of action—that is to say, a specific chemical compound responsible for their activity—has still not been identified. One might therefore speculate that their polyphenol content in general contributes to the unique effects. As already discussed in the broader context of food plants, a familiar issue arises once again for the phenols in medicinal plants: polyphenols intrinsically display a low level of bioavailability, are subject to chemical modification during processing, and are drastically modified by the metabolic activity of both intestinal flora and human enzymes. This is another field in which research is still in an embryonic state.
References
[6] A. Crozier et al., Dietary phenolics: chemistry, bioavailability and effects on health, Nat. Prod. Rep. 2009, 26, 1001–1043. https://doi.org/10.1039/b802662a
[7] Phenol Explorer database, phenol-explorer.eu.
[8] B. Cerda et al., Evaluation of bioavailability and metabolism in the rat of punicalagin, an antioxidant polyphenol from pomegranate juice, Eur. J. Nutrition 2003, 42, 18–28. https://doi.org/10.1007/s00394-003-0396-4
[9] A. Stalmach et al., Metabolite profiling of hydroxycinnamate derivatives in plasma and urine after the ingestion of coffee by humans: Identification of biomarkers of coffee consumption, Drug Metabol. Disp. 2009, 37, 1749–1758. https://doi.org/10.1124/dmd.109.028019
[10] N. Kuhnert, Unravelling the structure of black tea thearubigins, Arch. Biochem. Biophys. 2010, 501, 37–51. https://doi.org/10.1016/j.abb.2010.04.013
[11] M. Wichtl (Ed.), Teedrogen und Phytopharmaka (in German), Wissenschaftliche Verlagsgesellschaft Stuttgart, Germany, 1997. ISBN: 9783804723696
[12] T. Dingermannn et al., Schneider – Arzneidrogen (in German), Elsevier – Spektrum Akademischer Verlag, Heidelberg, Germany, 2004. ISBN: 9783827414816
The Author

Nikolai Kuhnert studied chemistry in Würzburg, Germany, and received his doctorate in 1995 under the supervision of Wolfdieter A. Schenk. After postdoctoral work at the Universities of Cambridge and Oxford, UK, he was appointed to his first academic position as Lecturer in Organic Chemistry at the University of Surrey, Guildford, UK. Since 2006, he has been Professor for Analytical and Organic Chemistry at Jacobs University, Bremen, Germany.
His research deals with the chemistry of plant polyphenols, especially from foods, with an emphasis on chemical modifications of plant phenols through food processing and metabolism, and consequences of the latter on human health.
The article has been published in German as:
- Polyphenole: Vielseitige Pflanzeninhaltsstoffe,
Nikolai Kuhnert,
Chem. unserer Zeit 2013, 47, 80–91.
DOI: 10.1002/ciuz.201300589
and was translated by W. E. Russey.
Polyphenols: Contributors to Good Health – Part 1
What are polyphenols and why do plants make them?
Polyphenols: Contributors to Good Health – Part 2
How do polyphenols influence human health?