Risks and Benefits of Herbal Remedies
The safety issues surrounding so-called herbal remedies have been called into question again and again. However, new regulations aimed at protecting consumers will not encompass all products in the way legislators and scientists might have hoped, as some products are classed as “food supplement” rather than medications. In light of this, Theo Raynor, Professor of Pharmacy Practice at the University of Leeds, UK, and colleagues have taken a close look at the safety information on herbal remedies. Their findings serve as a warning to people who turn to alternative medicine without first informing themselves of the risks and benefits.
More than one third of pharmaceutical products available have a natural product origin from Aspirin to Taxol including a myriad of antibiotics and anticancer agents. Indeed, some people feel that nature has somehow provided us with a medicine cabinet stocked with remedies for every ailment. But this is misguided if those people assume we can simply pluck a remedy off nature’s shelves and be cured.
Herbal Remedies Versus Pharmaceuticals
Despite the humble herbal origins of so many drugs, modern medicine is divided in many countries between the clinically tested and licensed products we know as pharmaceuticals and the alternative, herbal remedies. This division serves commercial interests well, specifically in the alternative medicine industry, with its claims of holistic approaches and natural cures. The herbal tag essentially allows the industry to avoid the years of trials and approvals required of the pharmaceutical industry. It does so by virtue of archaic laws that somehow equate natural with good and synthetic with bad, despite the fact that herbal often simply means impure and unproven.
 Legislation to improve quality control and labeling is gradually enveloping the herbal and alternative medicines so that the status of those with some proven efficacy might be raised to closer to that of conventional drugs. Those that fail to make the grade will, in many countries, be forced off the market. Until then, however, there is a need to consider the real dangers of herbal remedies.
Legislation to improve quality control and labeling is gradually enveloping the herbal and alternative medicines so that the status of those with some proven efficacy might be raised to closer to that of conventional drugs. Those that fail to make the grade will, in many countries, be forced off the market. Until then, however, there is a need to consider the real dangers of herbal remedies.
Many users and advocates claim that adverse drug reactions (ADRs) and side-effects are common with pharmaceutical products and that somehow the herbal products side-step this guilty secret of medicine. The reality is that side-effects are part of the risk-benefit equation that must be balanced when prescribing any medication, pharmaceutical or herbal. Herbal remedies are not immune to this balancing act.
Moreover, many people self-prescribe herbal remedies, so the side-effects and ADRs often remain hidden from official reports. Underlying conditions or conventional medicines taken in parallel become the point of blame for a deleterious outcome because the patient’s healthcare provider is unaware of any alternative medication being taken. Even if healthcare workers are aware, the nature of herbal remedies means that ingredients are not definitive, especially worrying in Traditional Chinese Medicine where ingredients vary wildly and even toxic heavy metals, such as arsenic and mercury, might be added.
Lack of Information
Raynor’s study, published July in the journal BMC Medicine, highlights the problem by showing how many over-the-counter herbal products fail to provide consumers with key information about the safe use of these products and the risks associated with them, especially when used in conjunction with prescribed medication.
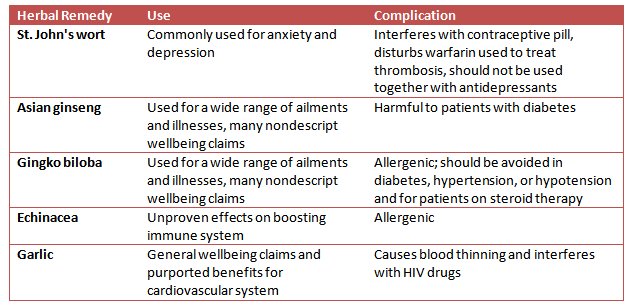
The team bought 68 different preparations of five commonly used remedies — St. John’s wort, Asian ginseng, echinacea, garlic, and ginkgo biloba — from health food shops, pharmacies and supermarkets across the UK. All of these remedies have known side-effects and contra-indications with other medications. They then compared the safety information provided with the products against data from the US National Center for Complementary and Alternative Medicine.
According to Raynor, “Consumers need reliable and comprehensive information when buying herbal remedies — information which tells them whether the remedy is suitable for them”. He and his colleagues found that most of the products (93 %) investigated were unlicensed. Since April 2011, EU rules require that herbal medicinal products be licensed or obtain a “Traditional Herbal Registration” (THR), which means the information with the product has been approved. In other words, the majority of remedies investigated did not meet safety or quality standards and more than half were being marketed as food supplements despite their known physiological or medicinal activity.
“The new European laws mean that herbal medicine products like St. John’s wort and echinacea should have complete and patient-friendly information about their safe use,” Raynor told ChemistryViews. “This contrasts, of course, with products like Ginkgo, Ginseng, and Garlic, which don’t make medical claims and will continue to be regarded as foods.”
Industry expert Sheryl Torr-Brown adds that, “Raynor’s findings are disturbing. It is difficult enough to predict side-effects with regulated drugs, let alone with herbal remedies that have unstandardized composition and no data to help a patient or practitioner manage potential risks. Surely, too, many of the serious outcomes of taking these agents go unreported because they also fall outside of current adverse event reporting regulations.”
Table 1. Herbal contra-indications
Claiming Efficacy While Avoiding Regulation
In the USA, the Food and Drug Administration (FDA) regulates supplements (which includes herbal remedies) as foods rather than drugs. In general, the laws about putting foods (including supplements) on the market and keeping them on the market are less strict than the laws for drugs. However, companies cannot make medical claims for their supplements without falling foul of the FDA.
“Herbal medicine companies cannot have their cake and eat it,” Torr-Brown told us. “All chemicals, natural or otherwise are toxic. It is the method of limiting our exposure to such chemicals, whether it be hemlock pulled from the woods, or a paracetamol (acetaminophen) from the shelf, that determines whether they provide benefit or harm.”
We should not allow alternative medicines to have the best of both worlds. The situation regarding herbal medicines is what Torr-Brown calls “A clear example of commercial interests outbalancing common sense and regard for consumer safety.” The herbal industry cannot claim efficacy and simultaneously avoid regulation. It is time to shake up nature’s medicine cabinet and restock the shelves, but only with those herbal remedies underpinned by science-based efficacy and safety information.
References
- Buyer beware? Does the information provided with herbal products available over the counter enable safe use?
D. K. T. Raynor, R. Dickinson, P. Knapp, A. F. Long, D. Nicolson,
BMC Medicine, 2011, 9(94).
DOI: 10.1186/1741-7015-9-94 - The analysis of heavy metals in Chinese herbal medicine by flow injection-mercury hydride system and graphite furnace atomic absorption spectrometry
X. Yuan, K. H. Ling, C. W. Keung,
Phytochem. Anal. 2009, 20, 293–297.
DOI: 10.1002/pca.1126 - Analytical aspects of drugs of natural origin
M. Hamburger, K. Hostettmann,
J. Pharm. Biomed. Anal. 1989, 7, 1337–1349.
DOI: 10.1016/0731-7085(89)80139-6
.




The UK has systematically limited herbal options for its citizens – all in the name of “science”. Big Pharma has grown geometrically while herbal suppliers have been chocked, stifled and trampled underfoot; all the while the UK suffers under the worst medical health system in “civilized” Europe. The UK nanny state can’t stand to see its subjects exercise any form of free choice be it: hospital, medicine, herbal supplements, type of fuel, means of garbage disposal, even down to how to fertilize the home garden. People of the UK you are citizens not subjects – revolt, resist defy and deny – throw off the yoke of your directed socialist economy – become free. Best Regards in your future endeavors. RJA