1. The Perfect (Boiled) Egg for Breakfast
Boiling the breakfast egg to perfection is far from child’s play. On the way from hen to breakfast table, so many complex chemical processes must take place that the outcome of the cooking phase cannot always be precisely predicted. It will, therefore, be worth our time to examine more closely the oological-chemical fundamentals of cooking eggs in order to avoid insofar as possible — and with the aid of solid scientific information — both disappointment and family crises around the breakfast table.

Figure 1. Soft-boiled egg in an egg cup, ready for breakfast, with typical (German) commercial markings.
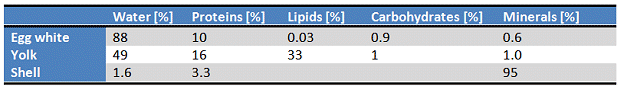
In many parts of the world, a freshly boiled (usually soft-boiled) egg is the crowning touch to every breakfast table, and is the very embodiment of a good start to the day. When we rap a knife on the white or brown shell of such an egg we are seldom conscious of what a technical marvel we are dealing with (Fig. 1). Under that unpretentious eggshell (which in itself is a structural masterpiece!) is concealed, in the narrowest of spaces, a fully-fledged culinary delicacy, one that deserves very close examination (see Tab. 1).
Table 1. A hen’s egg typically weighs about 60 g, has a surface area of 53 cm2, and consists of a shell 0.3–0.4 mm thick weighing about 6 g, 34 g of egg white, and 19 g of yolk. The individual components are made up as follows [1].
We will now delve into more details regarding egg production, and into typical national food regulations, using the case of Germany as an example (see: “Egg Output Data and Food Regulations Applicable in Germany“).
It is appropriate that we start at the ultimate source, the hen. It is convenient that there is absolutely no need for us here to confront that age-old question “Which came first, the chicken or the egg?”, because none of the eggs on the market will have been fertilized. They are thus utterly incapable of generating either hen or rooster.
2. In the Beginning Was the Hen
All modern chickens are thought to be descendants of the red junglefowl (Gallus gallus), and more specifically from the Javan subspecies (G. g. Bankiva; see Fig. 2), probably hybridized to some extent with the grey junglefowl. G. g. Bankiva still exists in the wild in Indonesian and Indian forests. Countless of these birds were carefully bred over the ages to improve their meat, or their ability to lay many eggs; some birds were also bred for show purposes, either because they were especially handsome, or because they were unusually aggressive, lending themselves to a very dubious form of entertainment.

Figure 2. A bankiva hen.
Characteristics prized in laying hens are their derivation from light- or medium-weight predecessors and they are now capable of producing as many as 280 eggs annually. One can only tip one’s hat to the breeders — and of course to the hens themselves! — considering that their bankiva hen ancestors typically laid only 8–12 eggs per year!
Production of a 60 g egg by a hen weighing roughly 1.7 kg represents an incredible biosynthetic feat. Such a laying achievement presupposes optimal environmental conditions and a diet rich in protein and calcium, one that can best be appreciated after translation into human terms. That is to say, one would need to imagine a person producing an egg weighing 2 kg (i.e., ca. 4.5 lb) almost every day, supported by an almost daily food intake of 4 kg (nearly 9 lb) of highest-quality nutrients!
Professor Waldemar Ternes, University of Veterinary Medicine Hannover, Germany, has suggested an even more dramatic comparison: the metabolic work accomplished by a hen in the course of ten days would correspond to a human being recreating his or her entire complement of arms and legs in the same time period.
3. The Composition of a Hen’s Egg
We now trace the path to the creation of a hen’s egg, with the associated phenomenal biosynthetic performances achieved by a laying hen [1–4] (see Fig. 3).
.jpg)
Figure 3. From ovary to egg. Modified schematic view based on Solomon [5].
t = 0 h: Ovulation
A hen’s ovary is the site where every 7–10 days there occurs maturation of one of the over 3000 egg cells (ova) initially present. Ovulation entails rupture of the thin follicle of such a cell, allowing the free ovum to drop into the infundibulum (see Fig. 3). At this point the ovum has a diameter of about 4 cm, and it embodies 30–33 % of the total weight of the eventual egg. The fat-in-water emulsion (water content ca. 50 %) of this “egg-to-be” already includes nearly all the fat and cholesterol found in a mature egg.
The vitellus or “yolk” contains, among other things, “yolk spheres”, 20–40 µm in diameter, whose structure is maintained even upon heating. Our tongue, an extraordinarily sensitive tactile organ, is able to feel out this substructure, which in turn is what confers upon the yolk of a hard-boiled egg its “mealy” character.
Lipovitellin, at 70 %, constitutes the chief lipoprotein of egg yolk; others include phosvitin (11 %) and what is known as the LDL or “low-density lipoprotein” fraction (12 %). Taken together the lipoproteins (66 %) lead to a yolk being the most fat-rich part of the egg as a whole.
The terms low- and high-density lipoprotein (LDL and HDL) here refer exclusively to the sizes of the lipoproteins themselves. In other words, this has nothing directly to do with so-called “bad” (LDL) and “good” (HDL) cholesterol, since the latter refer to proteins in our blood, ones to which cholesterol is bound.
Consumers often believe erroneously that a robust yellow color for an egg yolk is a measure of quality. In truth there is absolutely no relationship between yolk color and freshness, or general husbandry of the hens, nutritional value, or flavor. The yellow-orange dye present in egg yolk is not even synthesized by the hen, but rather is ingested directly or at least in the form of a precursor along with its food.
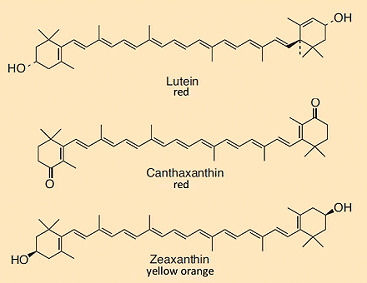
Figure 4. Carotinoids found in egg yolk.
The sources of the characteristic color of an egg yolk are orange and red carotinoids (see Fig. 4), typically mixed in along with the green mash the hens are fed. In the case of industrial egg production, carotinoids are added directly to standard mash. The effect is striking [6]: Figure 5 shows sections through 10 eggs, laid on successive days by the same hen after switching from a standard mash to one to which 20 mg/kg of carotinoids have been added. The color uptake, starting at the outer perimeter of the yolk, is clearly apparent with egg cells each of which has been allowed to mature for one day longer in the hen’s ovary.
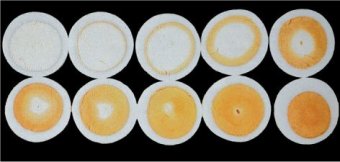
Figure 5. Cross-sections through 10 eggs from the same hen, laid on successive days after switching from a normal mash to one containing 20 mg/kg of carotinoids.
Figure 6 shows the effects of the reverse experiment: sections through 10 eggs laid on successive days by the same hen after switching from a mash supplemented with 20 mg/kg of carotinoids to one free of colorant.

Figure 6. Cross-sections through 10 eggs from the same hen, laid on successive days after switching from a mash containing 20 mg/kg of carotinoids to one free of coloring agents.
0 < t < 3 h: Formation of the Egg White
Shortly after the egg cell enters the oviduct, fertilization would become possible, unless it is prevented by the farmer, through a strict monastic separation of the sexes. Independent of whether fertilization occurs or not, every egg cell is converted within about 24 hours into a fully developed egg.
In the infundibulum, the funnel-shaped portion of the oviduct, egg albumen (egg white) is formed, along with its associated chalazae: a pair of spiral bands of protein within the egg white, twisted to the right, which extend out from the yolk at opposite ends of the egg and attach to the central viscous lining membrane of the egg white (see Fig. 7). These provide a “shock absorber” effect for the suspended yolk. Additional layers of egg white are added within the second zone of the oviduct, the magnum.
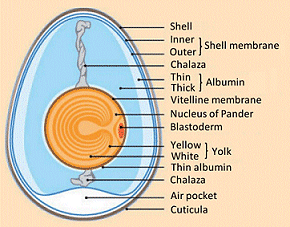
Figure 7. Schematic cross-section through a hen’s egg.
The inner and outer shell membranes, which have a total weight of only 140 mg, are formed in the part of the oviduct known as the isthmus. Calcium carbonate deposition commences within the short tubular section of the uterus known as the shell gland, beginning on the wart-like end portions of the outer membrane fibers.
The egg remains in the uterus “pocket” for a total of 20 hours, during which the rest of the shell forms — including the outer wall or cuticula.
The albumen, or egg white, constitutes ca. 60 % of an egg’s weight, and is a 10 % solution of various proteins (for details see: “Proteins Present in Egg White“). A distinction is made between four concentric layers of differing viscosity. The innermost protein layer (vitellin membrane) forms a solid protein sleeve around the yolk, terminating at the spiral (protein) chalazae bands. Their fixation in a highly viscous albumen layer laid down later guarantees an elastic suspension of the yolk in the egg white.
Outside of the vitellin membrane, and adjacent to it, are respectively three more albumen layers: One of low viscosity, one that is tougher, and then another with low viscosity. The differing viscosities of these layers is a function of the relative proportions of the roughly 40 dissolved proteins present. Overall, ovalbumin is the most prevalent (Fig. 8), representing ca. 60 % of the total protein content. It becomes denatured at the interface with air simply by vigorous beating, making it possible to prepare the stable whipped egg white that plays such a key role as the basis for meringues, soufflés, and angel food cake.
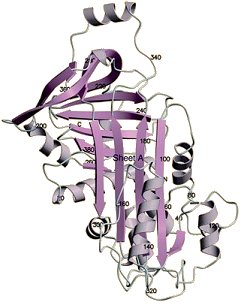
Figure 8. Crystal structure of ovalbumin.
Among the other “egg-white proteins”, perhaps the most important is ovomucin, for its function in establishing the layer-like structure of egg white. A high content of ovomucin causes the corresponding egg-white layer to become highly viscous, developing gel-like properties, since the protein itself has a tendency to aggregate and form filamentous structures.
3 < t < 4 h: Formation of the Inner and Outer Shell Membranes
The egg white is surrounded by two shell membranes, largely tightly bound together. The inner of the two has a thickness of 22 µm, the outer ca. 48 µm. Each consists of a closely woven network of protein filaments, acting together with the eggshell to protect the egg from penetration by bacteria. At the blunt end of the egg the two membranes separate, forming an air sac with a diameter of ca. 5 mm.
4 < t < 24 h: Formation of the Eggshell, Pigmentation, and the Cuticula
Calcium carbonate crystals are deposited on the matrix created by the protein filaments of the outer shell membrane. If one were to implant a ping-pong ball in a hen’s uterus, no shell formation would occur. On the other hand, were one to use a ping-pong ball to which outer shell membrane had been bonded, an “artificial” eggshell would form [10].
The contact between outer shell membrane and the innermost crystalline layer (Fig. 9) is made especially clear with the aid of images taken using a scanning electron microscope (see Figs. 10–12).
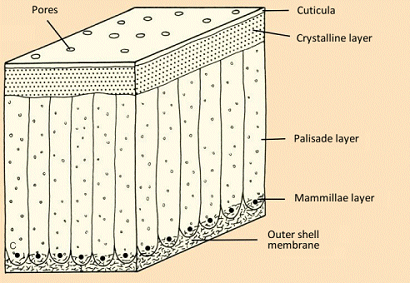
Figure 9. Eggshell structure.
During eggshell formation [11, 12], the first step is application of a three-dimensional network of delicate protein filaments. Calcium carbonate in the form of calcite crystallizes on this matrix until the intervening space has been filled (see Figs. 10 and 11). A second crystalline form of calcium carbonate, also found in nature, is aragonite, which is the basis of clamshells, pearls, and, for example, the shells of reptilian eggs.

Figure 10. Calcium carbonate deposition commences on the wart-like mammillen of the outer shell membrane (view from within, 50-fold magnification).

Figure 11. Calcite is deposited on the network of fine protein filaments from the outer shell membrane (view from within, 500-fold magnification), and crystallizes upwards in columnar form on an organic matrix (palisade layer).
Eggshell formation begins at the outer shell membrane with deposition of a mammillae layer ca. 70 µm thick, which transitions into the palisade layer. It is above this that the actual crystalline layer is to be found (see Figs. 9–12).

Figure 12. The outermost thin calcium carbonate layer is made of crystals oriented vertically, which are coated with a protein layer (cuticula) (external view, 50-fold magnification), such that the surface of the egg is smooth with a silky sheen. Pigments producing color are restricted to this uppermost calcium carbonate layer and the cuticula.
An eggshell is permeated by 7,000–17,000 pores with a diameter of 10–60 µm (see Fig. 13), which permit facile gas exchange with the surroundings, while at the same time serving as a barrier against penetration by dirt or microorganisms.

Figure 13. Pores at the shell surface (1500-fold magnification).
Only the outermost regions of the calcium layers of the shell are pigmented. The actual amount and color of the pigment introduced is established genetically. Hens that lay white eggs are Mediterranean varieties, such as leghorns. Those producing brown eggs are of Asiatic strains, including Rhode Island reds. A few strains even lay bluish or greenish eggs (see Fig. 14).
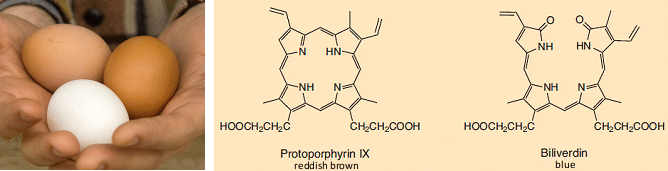
Figure 14. Eggshell pigments.
Araucanas (see Fig. 15) and certain other strains incorporate blue biliverdin or its green zinc complex. The actual pigments are produced in shell glands, and dispensed by papillae lining the oviduct [13].

Figure 15. An araucana hen. Photo credit: Willowbrook Park.
As suggested above, the amount, chemical composition, and distribution of pigments depends upon the strain of the hen, and the pigments themselves — protoporphyrin for brown and biliverdin-IX and its zinc complex for blue or green eggs — are synthesized in the shell glands [13,14].
Brown eggs are preferred by consumers — at least in some areas — who associate them with “natural, organic farming”. Actually, uniform brown pigmentation is simply a genetically derived attribute, since pigmentation serves in nature to protect the clutch of eggs, for which reason, the eggs of birds that lay their eggs on the ground are usually speckled [15,16]. In white eggs, pigment is almost entirely absent, but there is, in truth, absolutely no relationship between pigmentation and taste, style of flock management, or nurture of the laying hen.
Eggshells are covered with the cuticula, a thin layer composed of proteins, polysaccharides, and lipids that contributes to the silky sheen associated with eggs.
As far as the consumer is concerned, an eggshell amounts to nothing more than practical, disposable packaging. This of course fails to take account of the enormous effort on the part of the hen to prepare such an aesthetically pleasing object from marvelous composite materials. In the process, the hen is required, for a single egg, to excrete 5 g of calcium carbonate; for a human that would correspond to 170 g (more than one-third of a pound) of the substance: and that every day!
The inorganic chemistry involved in shell construction would appear to be trivial, since precipitation of calcium carbonate by simple exhalation through an aqueous solution of calcium hydroxide is one of the first examples of detection of an element one would encounter in a chemistry class.

As simple as this reaction would seem for us, a hen would disagree mightily. Indeed, crystallizing such an enormous amount of calcium carbonate leaves her completely out of breath. How so?
We need only to undertake a small calculation. The total CO2 reservoir in the body of a laying hen weighing 1.7 kg amounts to ca. 13 mMol. Producing a single egg shell requires 50 mMol. In other words, within a 24-hour period, her entire CO2 reservoir needs to be replaced almost three times. She is thus compelled to eat a great deal. The most problematic factor, however, is dealing with the resulting protons. The daily diet provides a hen, through metabolism, with roughly 5 Mol of protons, but this is precisely balanced out through a corresponding utilization (homeostasis) [17].
Were the hen not to take compensatory measures, the pH of her body fluids would fall below 1 (!!), clearly a lethal value! The hen combats this threat of over-acidification through increased release of carbon dioxide, whereby the pH of the blood rises. This stimulated metabolism in a hen is actually quite apparent: when there is no egg in her uterus she breathes during the night while asleep only about seven times per minute, but more like twelve times per minute when an egg is present [18]. In other words, from the standpoint of the hen, creation of a shell is in no way simply a passive act of waiting, but rather a heavy piece of work.
t = 24.2 h: The Laying Process
The hen lays her egg with the blunt end first. An egg assumes its typical form while still in the oviduct (Figs. 3 and 4). It is, therefore, not a function of the laying process. Topologically, an egg is a linearly deformed ellipsoid, an ovaloid, where the extent of the distortion is again a function of genetics.
Irrespective of how round or deformed the hen’s egg, we will continue to follow its path to the breakfast table. In Part 2 we’ll watch the egg going from hen to a pot on the stove, whereas Part 3 is devoted to experimental reproduction of the chemical highpoints of cooking an egg.
References
[1] W. Ternes, L. Acker, S. Scholtyssek, Ei und Eiprodukte, Paul Parey Verlag, Berlin, Germany, 1994. ISBN: 9783860224045
[2] W. J. Stadelman et al., Egg Science and Technology, 4th ed., The Haworth Press, Binghamton, USA, 1995. ISBN: 9781560228554
[3] W. Ternes, Naturwissenschaftliche Grundlagen der Lebensmittelzubereitung, Behr’s Verlag, Hamburg, Germany, 2008. ISBN: 9783925673849
[4] G. Lück, P. Gaymann, Eiweisheiten: Experimente rund ums Ei, Herder Spektrum, Freiburg, Germany, 2005. ISBN: 9783451054952
[5] S. E. Solomon, Egg and Eggshell Quality, Manson Publishing, London, UK, 1997. ISBN: 9780723416470
[6] A. Hatzipanagiotou, W. Hartfiel, Arch. Geflügelkde. 1984, 48, 155.
[10] G. Krampitz et al., Berl. Münch.Tierärztl. Wschr. 1973, 86, 313. Link
[11] Y. Nys et al., Poultry Avian Biol. Rev. 1999, 10, 143.
[12] I. Lavelin et al., Poultry Sci. 2000, 79, 1014. Link
[13] R. Zhao et al., Poultry Sci. 2006, 85, 546. Link
[14] J.-H. Fuhrhop et al., Liebigs. Ann. Chem. 1975, 1131. DOI: 10.1002/jlac.197519750612
[15] T. K. With, Biochem. J. 1974, 137, 596. Link
[16] R. M. Kilner, Biol. Rev. 2006, 81, 383. DOI: 10.1017/S1464793106007044
[17] K. G. M. M. Alberti et al. The Hydrogen Ion in Normal Metabolism: A Review in Ciba Foundation Symposium 87 – Metabolic Acidosis (Eds: R. Porter, G. Lawrenson), John Wiley & Sons, Chichester, UK 1982. DOI: 10.1002/9780470720691
[18] P. Mongin, World’s Poultry Sci. J. 1968, 24, 200. DOI:10.1079/WPS19680021
Author
Prof. Klaus Roth
Freie Universität Berlin, Germany.
The article has been published in German in:
and was translated by W. E. Russey.
Boiled Eggs: Soft and Hard — Part 2
In Part 2 we’ll watch the egg going from hen to a pot on the stove
Boiled Eggs: Soft and Hard — Part 3
Part 3 is devoted to experimental reproduction of the chemical highpoints of cooking an egg
Boiled Eggs: Soft and Hard — Part 4
In Part 4, we reach the practical part as we discover the ideal egg, from a culinary perspective
Other articles by Klaus Roth published by ChemViews magazine:
- In Espresso — A Three-Step Preparation
Klaus Roth proves that no culinary masterpiece can be achieved without a basic knowledge of chemistry
DOI: 10.1002/chemv.201000003 - In Chocolate — The Noblest Polymorphism
Klaus Roth proves only chemistry is able to produce such a celestial pleasure
DOI: 10.1002/chemv.201000021 - In Sparkling Wine, Champagne & Co
Klaus Roth shows that only chemistry can be this tingling
DOI: 10.1002/chemv.201000047 - In Chemistry of a Hangover — Alcohol and its Consequences
Klaus Roth asks how can a tiny molecule like ethanol be at the root of so much human misery?
DOI: 10.1002/chemv.201000074 - In The Chemist’s Fear of the Fugu
Klaus Roth shows that the chemist’s fear of the fugu or pufferfish extends as far as the distinctive and intriguing poison it carries
DOI: 10.1002/chemv.201000104 - In Chemistry of a Christmas Candle
Klaus Roth explains that when we light a candle, the chemistry we are pursuing is not only especially beautiful, but also especially complex
DOI: 10.1002/chemv.201000133 - In Pesto — Mediterranean Biochemistry
Klaus Roth uncovers the nature of this culinary-chemical marvel, and thereby comes to enjoy it all the more
DOI: 10.1002/chemv.201200001 - Video Interview with Klaus Roth



