Spin is a fundamental property of atoms and molecules. The respective chemistries of technologically critical transition metals in the d- and f- blocks of the periodic table are especially sensitive to spin. It directly determines or strongly influences numerous physio-chemical phenomena found in enzymatic synthesis, spin-crossover compounds, metal-oxo complexes, catalysis, photophysical properties, transitions, etc. In a recent perspective article published in the International Journal of Quantum Chemistry, Marcel Swart, ICREA Research Professor at University of Girona, Spain, discusses recent advances in calculation of spin-state splittings of transition-metal complexes, with concrete examples and a view towards future work in the field.
Modeling of spin states has historically proven to be particularly difficult. Having started some work on cytochrome P450 enzymes during his postdoc, Professor Swart details some of the challenges, “This catalytic cycle is one of the most intriguing mechanisms I have seen so far, as it contains several spin flips, an electron transfer that only takes place when the substrate has entered the active site, and a dioxygen that enters only after the electron transfer has taken place. The steps leading to product formation are still not completely understood, after nearly fifty years of research on them.” In addition, the sensitivity of first-principle calculations to the level of theory, the quality of the basis set, and other factors like the inaccuracies in traditional and popular density functional theory (DFT) functionals make theoretical spin-sensitive studies difficult. The author has reviewed recent contributions using several different applications as illustrative examples. Two of these applications with far-reaching consequences are metal-oxo complexes and spin-crossover compounds.
Metal-oxo Complexes
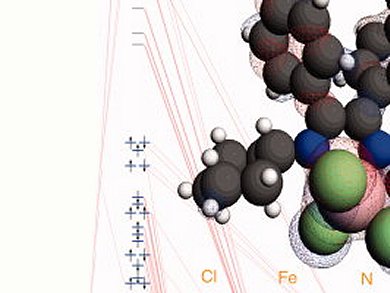
Metal-oxo complexes are relevant in many aspects of electron transfer, oxygen transfer (such as heme-containing proteins in blood), gene-regulation, etc. These have important implications for our understanding of the functioning of various biological systems. Various transitional metals, such as Zn, Cu, Fe, play an important role in the regulation of various metabolic processes in the human body. “One of the interesting peculiarities of these metal-oxo complexes is that within enzymes they are mostly observed in a high spin-state, while in biomimetic complexes they are mainly intermediate spin. Without any doubt, the enzyme environment plays a large role here, which makes them very interesting systems to investigate”, says Swart.
Metal-oxo complexes can be understood by using exchange-enhanced reactivity (EER) following work of Sason Shaik and colleagues, The Hebrew University of Jerusalem, Israel. The EER principle recognizes that exchange interactions on the metal may change during a reaction, which has the dual effect of changing the reaction barrier and complicating the numerical calculations necessary. Professor Swart underscores the importance of this advance by saying, “The biggest advancement for the community would be the understanding of which methods one can use without any problem, and which ones to avoid. With this knowledge, the community could move on to study the chemistry of transition-metal complexes, such as for instance, the catalytic cycle of P450 enzymes. This is the final goal of our research, understanding chemistry and reactivity patterns. The exchange-enhanced reactivity by Shaik and co-workers is one of the most important developments in this respect.”
Spin-Crossover Compounds
Spin-crossover (SCO) compounds exhibit changes in spin-states in response to temperature, pressure, etc. This behavior makes them excellent candidates for novel electronic and magnetic devices that may offer new functionalities for use in sensors, complex circuit elements, and other such applications. The author’s studies using non-standard functionals provide good agreement between computed spin-state splittings at 0 K and experimentally observed SCO-behavior. With the adaptation of ligand-field molecular mechanics (LFMM) methods, in-silico design of SCO (or analogous light-induced excited spin state trapped – LIESST) has become possible.
Building a Consensus
The perspective recognizes that this is a very fluid field with new contributions coming to light rapidly and recommends the use of a combination of approaches to serve as sanity check in various calculations. According to Professor Swart, standard density functionals (B3LYP, BP86) cannot be trusted for spin state energies, and the selection of basis set can play a major role in the results. Right now a number of new density functionals have been developed that seem to account correctly for the spin state splittings for some systems, but it is not yet clear if any of them works well for all systems and can be used to draw any conclusions about general trends.
The formation of scientific consensus on which methods can be trusted and which ones should be avoided for the simulation of spin-state is a fundamental, and an ongoing process; “This is why I’m organizing a CECAM workshop in Zaragoza, Spain, from September 18–21, 2012, where both experimentalists and theoreticians will be present,” Professor Swart concludes.
As the field develops, quantum chemists hope to provide greater insights into areas as diverse as magnetic devices and metabolic processes in the human body.
- Spin states of (bio)inorganic systems: Successes and pitfalls,

Marcel Swart,
Int. J. Quantum Chem. 2012.
DOI: 10.1002/qua.24255
Other articles available free to read at http://reviews.q-chem.org/