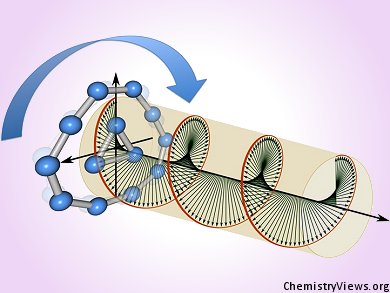
Molecular Rotary Engine
Shining circularly polarized infrared light on a boron cluster makes an inner ring of atoms turn within an outer ring of atoms like the piston in a rotary internal combustion, or Wankel, engine. Nature exploits molecular motors endlessly in the flagella of bacteria, sperm tails, and the ATP powering of every cell, but chemists have observed these biological nanomachines in awe hoping that one day they might emulate their tiny moving parts in a molecular machine of their own.
According to Anastassia Alexandrova, University of California, Los Angeles, USA, and colleagues both there and at Utah State University, USA, clusters of atoms, which lie beyond the atomic scale but are not considered to be simply fragments of a bulk solid, might offer chemists one entry point for creating their own molecular machines. As such, Alexandrova and colleagues have turned to boron, well known for its cluster-forming abilities, to build a nanoscale device that resembles the Wankel engine.
In said engine, a three-faced piston sits within a cylinder and is made to rotate through sequential combustion of fuel within each cylinder formed by the face of the piston and the curved inner wall of the cylinder. Unlike the more familiar piston engine in which pistons move up and down, the energy of combustion is converted directly into rotation of a drive shaft rather than the up and down linear motion of pistons which then needs to be converted into rotation via a system of cams.
Alexandrova’s molecular Wankel engine is driven by light rather than combustion and sees the inner piston formed of a three-atom boron cluster rotating relative to a ten-atom “cylinder”. The piston rotates at 300 gigahertz, 18 trillion revolutions per minute, rpm, somewhat higher than the several thousand rpm of vehicular Wankel engines.
Controlling the Direction of Rotation
The team had previously demonstrated the free movement of a ring-like boron cluster within a larger cylinder in which the energy barrier to relative rotation of each was rather low. In this scenario the piston was free to rotate in either direction. Now, the team has demonstrated that an infrared laser at 3 terahertz can drive a smaller piston to rotate in one direction only relative to its cylinder. The team explains that the rotation involves 30 sequential movements of the inner ring, the piston, relative to the outer ring, the cylinder of their B13+ cluster. The team points out that an applied electric field raises the barrier to counterclockwise rotation ensuring that the engine is essentially always driven in the same direction.
Each elementary rotation generates about 2 kilojoules per mole; this energy is easily dissipated to a thermal bath at 300 Kelvin without raising the temperature of the molecular engine. Other attempts to build molecular-scale motors, driven by chemical energy or an electric current, have often been stymied by excess heat and the disturbances to the structure that result.
“In order for a device like this to be employed, it needs to be suspended in some kind of framework, like a metal-organic framework (MOF), or put on a supporting surface that does not bind it too strongly, perhaps just via weak van der Waals interactions, or otherwise be secured and aligned with other clusters in the ensemble,” explains Alexandrova. “Such a device might look like a tiny solar panel, except up-taking terahertz radiation.”
“This result is a breakthrough in new boron chemistry,” says Alexander Boldyrev, Utah State University, a computational chemist with a particular interest in nanotechnology and boron clusters. “Taking into account the recent syntheses of new solid materials containing planar boron clusters, I hope that clusters like B13+ and B19– might become the building blocks of yet to be synthesized solids.”
Image: © Wiley-VCH
- B13+: A Photodriven Molecular Wankel Engine,
Jin Zhang, Alina P. Sergeeva, Manuel Sparta, Anastassia N. Alexandrova,
Angew. Chem. Int. Ed. 2012.
DOI: 10.1002/anie.201202674