The Challenge of Bonding Wet Surfaces
When gluing things together, both surfaces usually need to be dry. Gluing wet surfaces or surfaces under water is a challenge. In the journal Angewandte Chemie, Korean scientists have now introduced a completely new concept. They were able to achieve reversible underwater adhesion by using supramolecular “velcro”.
Previously, researchers have primarily attempted to mimic natural underwater “adhesives” like the adhesive molecules used by mussels. Such substances adhere well in wet surroundings, but the adhesion is not reversible. A team led by Kimoon Kim at Pohang University of Science and Technology in Korea has chosen a different approach: they have moved away from natural models—and achieved reversible underwater adhesion.
The primary challenge for conventional adhesion under water is that the water molecules between the surfaces to be glued and the adhesive need to be removed so that these surfaces can react chemically. This is not the case with the new approach. Kim and his co-workers use host–guest interactions between water-soluble host molecule with a hydrophobic pocket and ionic guest molecule with a hydrophobic block. They form robust noncovalent bonds in water. In fact, the repulsion of water molecules is the driving force for formation of the bond, mediated by ion–dipole and hydrophobic interactions.
Ferrocene and Curcurbit[7]uril Host–Guest Interactions
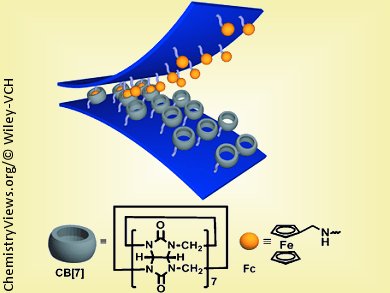
The researchers chose curcurbit[7]uril as their host and aminomethylferrocene as the guest. Cucurbit[n]uril (n = 5,6,7,8 and 10) are macrocycles—large, ring-shaped molecules made of multiple glycoluril units that can bind other molecules within their cavities. Their name is derived from cucurbita, the Latin name for pumpkin, which they resemble in shape. Ferrocenes are also known as sandwich compounds. The “bread” consists of two aromatic rings of five carbon atoms, while the “filling” is a single iron atom held in the middle. The ferrocene chosen by the researchers lodges very securely and specifically inside a “pumpkin” made of seven glycoluril units.
The researchers produced some silicon strips with many “pumpkins” attached, as well as some with many “sandwiches”. When these strips come into contact with each other they stick together tightly, similar to Velcro. Once stuck together, a 1 x 1 cm piece of this supramolecular velcro can hold a weight of 2 kg in water. After drying in air it can hold as much as 4 kg. This is more than double-sided sticky tape can hold.
Like a macroscopic Velcro strip, the molecular version can be separated with a strong pull and reused multiple times. The adhesion can also be reversed chemically through application of a hypochlorite solution, which oxidizes the iron atoms. After reduction with an agent such as ascorbic acid, the velcro can adhere again.
Because the materials used are biocompatible, biological applications may be possible, for example in surgery suture or repairing live tissue. In comparison, the mussel mimetic underwater adhesives require strong oxidizing agents for curing to these applications, which is not the case with the new system.
- Supramolecular Velcro for Reversible Underwater Adhesion,
Youngjoo Ahn, Yoonjung Jang, Narayanan Selvapalam, Gyeongwon Yun, Kimoon Kim,
Angew. Chem. Int. Ed. 2013.
DOI: 10.1002/anie.201209382