Alzheimer’s Treatment
Light-activation of the well known xanthene dye, rose Bengal, might lead to new insights and approaches to halting the aggregation of errant amyloid proteins in the brain which are closely associated with the lethal neurodegenerative disorder Alzheimer’s disease (AD). Researchers from South Korea will report details of the findings in the January 2015 issue of the journal Biomaterials.
It has been said many times, but as living standards, medical advances and good nutrition improve, so populations are aging as more and more people survive the trials and tribulations of infancy, youth, young adulthood and middle age and anticipate a long and healthy retirement. Unfortunately, having survived lethal infectious diseases and other killers in one’s younger days does not protect against the diseases that ravage one’s old age – cardiovascular disease, stroke and heart attack, and neurodegenerative diseases such as Parkinson’s and Alzheimer’s. As such, an aging population means an increase in the numbers of people who will succumb to such diseases applying the pressure of medical urgency on medicinal chemists and others to find and develop novel agents that might inhibit the degradation or block other causative pathways in such diseases.
Joon Seok Lee, Byung Il Lee, and Chan Beum Park of the Korea Advanced Institute of Science and Technology (KAIST), in Daejeon, point out that 13 % of the population over the age of 65 years is affected by AD, making it the most common progressive brain disease. There are two main schools of thought regarding the cause and progression of this terminal dementia that lays waste to memory and cognition. The first hypothesis, on which most available drugs are based, is the cholinergic hypothesis. This suggests that AD is caused by reduced synthesis of the neurotransmitter acetylcholine. However, the pharmaceuticals aimed at treating AD rarely have much effect and there is an increasing recognition that while the biochemistry may be associated with the disease, the hypothesis is no longer considered entirely valid by the research community. A second hypothesis, first postulated in 1991, seems to sit on much firmer ground.
Amyloid Hypothesis
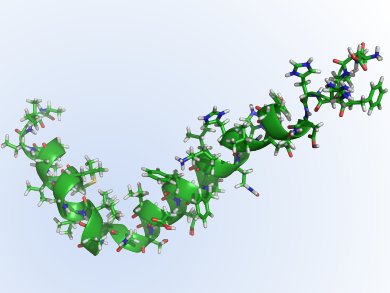
The amyloid hypothesis says that deposits of malformed protein, amyloid-β (pictured), form toxic aggregates, so-called plaques, which accumulate in the brain and damage neurones and give rise to the increasingly and worryingly familiar symptoms of dementia. Post mortem evidence of patients supports the case for the amyloid hypothesis as does genetic evidence from amyloid precursor protein and the association with chromosome 21 in people with Down syndrome.
Chemically speaking, monomeric amyloid-β is a polypeptide of 39 to 42 amino acids, released following sequential proteolytic cleavage of β-amyloid precursor proteins by β- and γ-secretase enzymes. Nucleation dependent polymerization of these strands gives rise to beta-sheet-like structures and fibrous aggregates. Since researchers first mooted the amyloid hypothesis, the idea that inhibiting its production and/or blocking its aggregation might slow or halt the development of AD.
Rose Bengal Blocks Aggregation of Amyloid-β
Park and colleagues have focused on a particular, well-studied group of compounds familiar to the biochemical and biomedical laboratory – the xanthene dyes. These compounds, which have a dibenzo[a,e]pyran 10H-9-oxaanthracene skeleton, include the likes of fluorescein, eosins, rhodamines. One of the fluoresceins, rose Bengal, 4,5,6,7-tetrachloro-3′,6′-dihydroxy-2′,4′,5′,7′-tetraiodo-3H-spiro[isobenzofuran-1,9′-xanthen]-3-one, is commonly used in optometry to stain conjunctival and corneal cells to highlight damage to those eye tissues. It – like other xanthenes – is also used widely as a stain for biological samples and live cells.
The team has now demonstrated that under green LED illumination rose Bengal can block the aggregation of amyloid-β. It has a high affinity for the monomeric form, much more so than any other xanthene dye they screened; fluorescein, eosin B, rhodamine B, and sulforhodamine B had negligible effects. The researchers suggest that light activation of this compound via laser-coupled optical fibers might provide the means to cleanly block the neurotoxic formation of amyloid-β plaques early in the diagnosis of AD patients.
“We observed significant red shift and strong enhancement of fluorescence emission of rose Bengal in the presence of amyloid-β-42,” the team reports. “Rose Bengal strongly inhibits aggregation upon its absorption of visible light energy (2.06 to approximately 2.75 electronvolts).
Piotr Hanczyc of Chalmers University of Technology in Sweden is developing a laser-based approach to treating pathogenic protein aggregation of the kind seen in AD, Parkinson’s disease, and Creutzfeldt-Jakob disease. “With any drug treatment of brain diseases there are two major problems: drug delivery through the blood-brain barrier and selective binding to amyloid with further possible drug activation,” Hanczyc explains. “Light activation is an interesting and promising approach whereas in this paper the authors selected some chromophores that can inhibit amyloid growth. Thus as models these molecules are interesting for developing photo-activated drugs,” he adds.
- Photo-induced inhibition of Alzheimer’s β-amyloid aggregation in vitro by rose Bengal,
Joon Seok Lee, Byung Il Lee, Chan Beum Park,
Biomaterials 2015, 38, 43–49.
DOI: 10.1016/j.biomaterials.2014.10.058