The Many Uses of MOFs
A new class of porous material based on a metal-organic framework (MOF) could double up as a hydrogen storage medium for vehicle fuel cells and as a sponge for the greenhouse gas carbon dioxide, according to research by US scientists.
Writing in the September issue of the journal Nature Chemistry, Joseph Hupp and colleagues at Northwestern University, Evanston, Illinois, explain how MOFs are hybrid porous materials comprising a two- or three-dimensional network of metal ions or clusters connected via coordinating organic bridges.
The unique properties of MOFs, not least their very high internal surface area has led to their investigation in gas purification and separation, as novel catalysts, as drug-delivery agents and in sensor technology. It is the possibility of storing large volumes of gas, essentially in a low-volume solid that has suggested the possibility of using MOFs as a safe and portable storage medium for hydrogen gas, the feedstock for electricity-generating fuel cells for “non-polluting” vehicles and portable devices.
In addition, the sequestration of carbon dioxide from the atmosphere, or from the vented exhaust gases of power generation plants that burn fossil fuels also offers an intriguing avenue of research into MOF materials.
Theoretical Structure
Hupp and colleagues realized that there are many different theoretical structures for solid MOFs based on any number of metals in combination with countless organic ligands. One might suppose that the vast range of putative building blocks available would mean that it would be quite straightforward to synthesize a MOF with fine-tuned properties almost on demand. This has not proven to be the case, although several useful MOFs have emerged from various laboratories around the world.
A particular issue that would-be MOF makers face is that removing the solvent from the pores commonly results in the collapse of the framework. With this historical perspective in mind, the Northwestern team turned to a theoretical approach to design their novel MOF.
An important aspect of MOF chemistry is that the products are crystalline, which means X-ray diffraction studies can be wielded to obtain precise molecular structures that can be fed into a computer model and the static and dynamic interactions of guests, such as small gas molecules, calculated. By tweaking the structure in the model it is then possible to pull out adsorption isotherms, binding energies and transport behavior from the theoretical pores and to predict what might happen if different building blocks are used in a synthesis.
From Model to Made
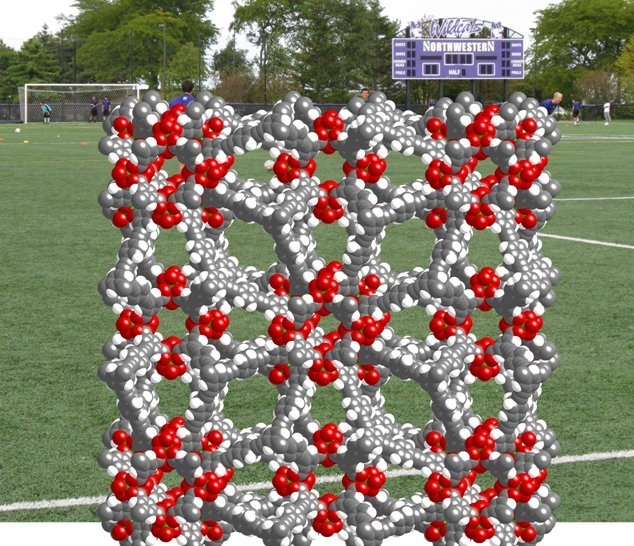
Team members Özgür Yazaydin and Randall Snurr explain that computational modeling has now allowed them to design and characterize in advance a MOF, which they refer to as NU-100 (also known as PCN-610), with a so-called (3,24)-paddlewheel connected network based on copper(II). This structure turns out to have a particularly high surface area to volume ratio. Experimental synthesis of the compound then led to a material with a structure matching the calculations and a surface area of more than 6000 m2/g. “1 gram of NU-100 has an internal surface exceeding the surface area of a football field,” Hupp told ChemViews, “Equivalently, one pound of NU-100 has an internal surface area of 1 square mile.”
They have tested how well the material absorbs hydrogen gas (164 milligram per gram) and carbon dioxide (more than 2300 milligram per gram). These figures also corroborated the modeling predictions, which bodes well for using the same theoretical approach to design other useful MOFs with specific selectivities and characteristics.
An area the size of a football pitch within a porous MOF could be used to store hydrogen or trap carbon dioxide (Credit: Farha et al.)
- De novo synthesis of a metal-organic framework material featuring ultrahigh surface area and gas storage capacities
O. K. Farha, A. Ö. Yazaydin, I. Eryazici, C. D. Malliakas, B. G. Hauser et al.,
Nat. Chem. 2010.
DOI: 10.1038/NCHEM.834