Converting Light to Motion
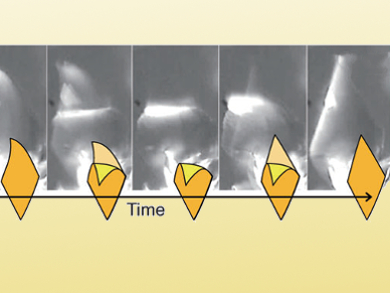
Microrobots and nanomachines are no longer far-off dreams. However, one of the biggest challenges that remains is to transmit the motion of individual molecules or molecular clusters into structured macroscopic motion that continues as long as the system is supplied with energy. In the journal Angewandte Chemie, Japanese scientists have now introduced such an autonomously oscillating system: A plate-like crystal that continuously alternates between bending and stretching as long as it is irradiated with blue light in water.
The platelets developed by a team led by Sadamu Takeda and Yoshiyuki Kageyama at Hokkaido University, Sapporo, Japan, are crystals containing a mixture of oleic acid and a derivative of azobenzene in a specific ratio. The critical component is two phenyl rings in the azobenzene compound that are bound together by an azo bridge (–N=N–).
Crystal Bends and Stretches in Blue Light
The rings can be in either a cis or trans orientation relative to each other, meaning that they can be either on the same side or on opposite sides of the imaginary plane containing the azo bridge. In the flat crystal, 99.8 % of the molecules are in the trans configuration. Blue light excites the molecules and the azo bridges “loosen up”, allowing some of the molecules to rearrange (isomerization) and take on the cis configuration. This changes the shape of the molecules, disrupts the crystal structure, and causes strain. Once a certain percentage of the molecules are converted to the cis isomer, the strain becomes so great that the morphology changes: The platelet bends.
If the irradiation continues, the platelet eventually flicks back to its original flat state. Why does this happen? The cis isomers are also excited by blue light and can change their structure – to the trans configuration. Exactly why the population of cis isomer starts to decrease is not fully understood. It is possible that the cis form of the molecule is better able to absorb the light energy; it is also possible that the changed molecular arrangement in the new crystalline phase changes the light absorption of the molecules relative to the “normal” crystal.
Macroscopic Self-Oscillation
Once the proportion of the cis isomer falls below the limiting level, the original crystal structure is resumed and the platelet flips back to its stretched conformation. The cycle then begins again. While the degree of bending is only determined by the dimensions of the individual crystals, the intensity of the light influences the rate of flipping. The stronger the irradiation, the faster the shape change.
This conversion of light energy to mechanical motion may be useful for the design of materials that mimic the movement of animals, cellular components, or dynamic technical components for applications such as micromachines.
- Dissipative and Autonomous Square-Wave Self-Oscillation of a Macroscopic Hybrid Self-Assembly under Continuous Light Irradiation,
Tomonori Ikegami, Yoshiyuki Kageyama, Kazuma Obara, Sadamu Takeda,
Angew. Chem. Int. Ed. 2016.
DOI: 10.1002/anie.201600218