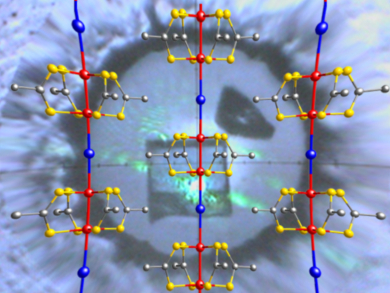
One-dimensional (1D) coordination polymers containing mixed-valent metals are expected to have outstanding electronic properties. 1D halogen-bridged mixed-valent transition-metal complexes – so-called MX- and MMX-chain families – have isolated one-dimensional d-electronic systems. In particular, some members of the MMX-chain family have high conductivity. Conductivity ranges from semiconductivity to metallic conductivity and originates from a binuclear metal unit that provides internal electronic flexibility. A representative compound, Pt2(dta)4I (dta: CH3CS2–), is known to have high electrical conductivity.
Kazuya Otsubo and Hiroshi Kitagawa, Kyoto University, Japan, and colleagues have investigated the pressure dependence of the conductivity and lattice parameters of Pt2(dta)4I. They studied the compound’s conductivity and its phase diagram using a combination of Raman spectroscopy, X-ray oscillation photography, and X-ray crystallography.
The researchers demonstrated that this compound shows semiconducting behavior under pressure with no significant electronic phase transition. Comparison with an isostructural compound, Pt2(dtp)4I (dtp: C2H5CS2–), showed that the length of the alkyl chain on the terminal ligand is important in determining the electronic states and hence the conductivity.
- Conducting Behavior and Valence Ordering of a One-Dimensional MMX-Type Coordination Polymer under High Pressure,
Kazuya Otsubo, Tomohiko Suto, Atsushi Kobayashi, Ryuichi Ikeda, Masato Hedo, Yoshiya Uwatoko, Hiroshi Kitagawa,
Eur. J. Inorg. Chem. 2016.
DOI: 10.1002/ejic.201600499