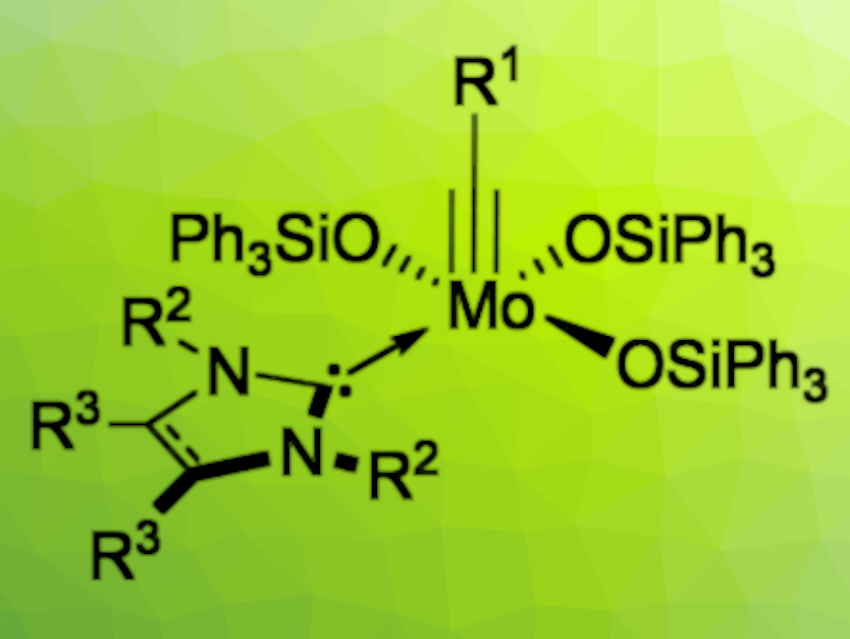
The development of air-stable catalysts for alkyne metathesis is an interesting research target for synthetic organometallic chemists. Ideally, the catalysts should be based on cheap and abundant metals (e.g., molybdenum or tungsten), not require the addition of chemicals for activation, and consist of well-defined, isolable species.
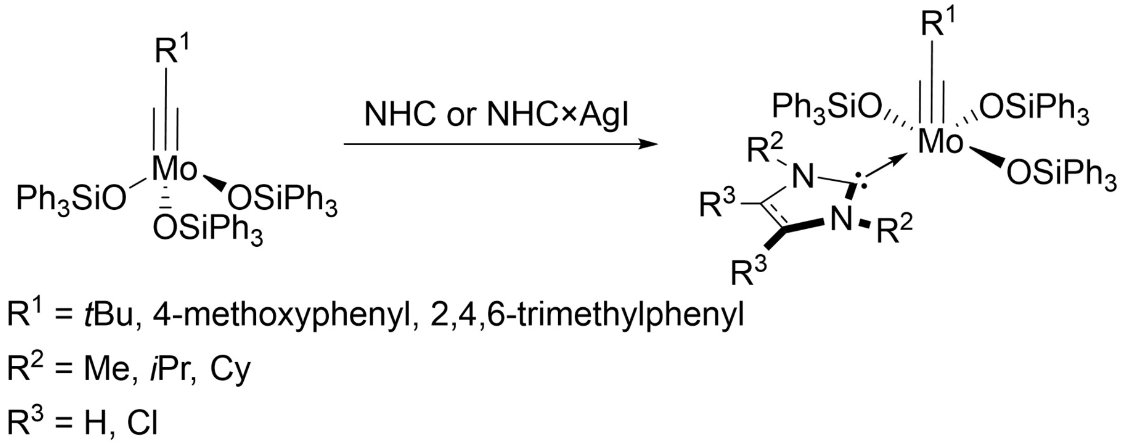
Michael R. Buchmeiser, University of Stuttgart, Germany, and German Institutes of Textile and Fiber Research (DITF), Denkendorf, Germany, and colleagues have developed catalysts that meet those criteria: a series of molybdenum alkylidyne silyloxy complexes that have an N-heterocyclic carbene (NHC) ligand coordinated to their vacant binding site (general structure pictured above). The complexes were synthesized from molybdenum silyloxy precursors of the type [Mo(≡C(R))(OSiPh3)3], either via a reaction with a free NHC in benzene or via a transmetallation with an NHC silver iodide adduct in CH2Cl2 (pictured below).
The prepared complexes show remarkable stability towards moisture and air. The most stable complex only shows slight signs of decomposition after exposure to the ambient atmosphere for one week. The novel complexes display excellent productivity (turnover number up to 6,000) in alkyne metathesis at 80 °C with substrates bearing functional groups such as tosyl, ether, ester, thioether, and nitro groups. Mechanistic studies suggest that at elevated temperatures, either the NHC or the silyloxy ligand dissociates and the active species is formed.
- Molybdenum Alkylidyne Silyloxy N‐Heterocyclic Carbene Complexes – Highly Active Alkyne Metathesis Catalysts that can be Handled in Air,
Janis V. Musso, Vincent Gramm, Sarjano Stein, Wolfgang Frey, Michael R. Buchmeiser,
Eur. J. Inorg. Chem. 2022.
https://doi.org/10.1002/ejic.202200649