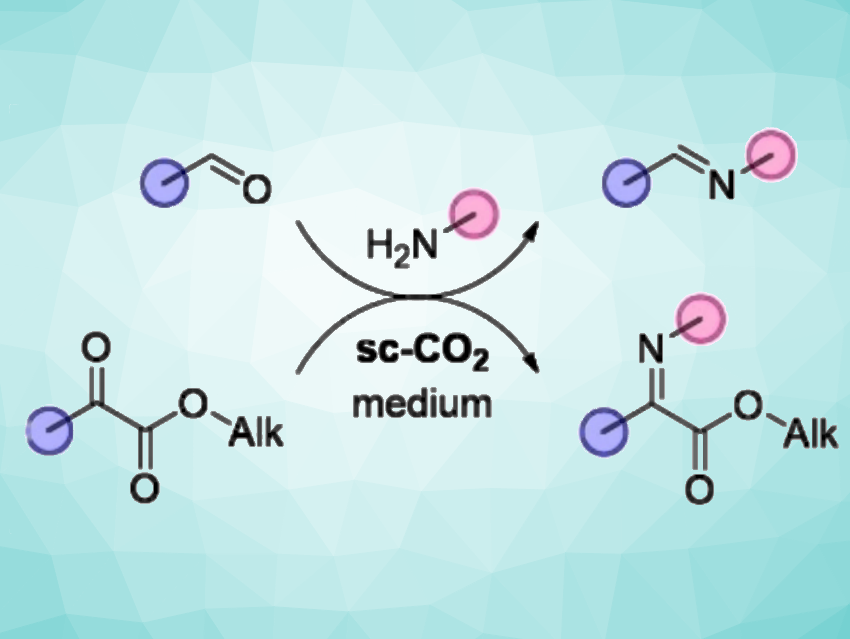
The environmentally friendly production of chemicals requires the elimination of toxic and harmful solvents from the processes. Imines are useful, e.g., as dyes, ligands, and intermediates for organic synthesis. Generally, imines are synthesized via the condensation of amines with carbonyl compounds or their precursors in organic solvents. These reversible reactions are commonly performed at high temperatures and often require catalysts (metals, acids, or bases). Thus, the development of new, environmentally benign production processes for imines would be useful.
Sergei Zlotin, N. D. Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences, Moscow, and colleagues have developed a convenient protocol for the “green” synthesis of various aldimines and ketimines in supercritical carbon dioxide (sc-CO2). Carbon dioxide is easily available, inexpensive, non-flammable, and non-toxic. In this condensation reaction, it acts both as a traceless, readily removable solvent and as a promoter.
The team performed the reactions of aldehydes with amines in sc-CO2 at 15 MPa and 35 °C. Various benzaldehyde derivatives were reacted with anisidine to give the corresponding imines in almost quantitative yields. Sterically hindered tert-butyl aldehyde gave lower yields. For the amine partner, other, less nucleophilic aniline derivatives gave moderate yields. The team also used the approach to convert α-ketoesters into the corresponding ketimines 8 MPa and 55 °C with longer reaction times (18 h).
The team proposes that the reaction in CO2 is autocatalytic: The water released during the condensation process forms carbonic acid with CO2, which accelerates the reaction. With the developed approach, the imine products were formed in high yields without conventional solvents. They were obtained in crystalline form and did not require further purification.
- Autocatalytic Green Synthesis of Imines in Traceless Medium,
Roman Ivanov, Elizaveta Ivanova, Vladislav Merkulov, Mikhail Zharkov, Ilya Kuchurov, Sergei Zlotin,
Eur. J. Org. Chem. 2023.
https://doi.org/10.1002/ejoc.202300366