The storage of solar energy is important to improve the utilization of renewable energy. Molecular solar thermal energy storage (MOST) systems can convert solar energy into chemical energy, store it, and later release it again. Certain molecules can isomerize under sunlight into a higher-energy isomer, and the stored energy can later be released as heat in a specifically triggered back-reaction.
The photo-induced [2+2] cycloaddition-cycloreversion equilibrium between norbornadiene and quadricyclane, for example, is useful in this context. The system has a high energy storage potential, and the energy can be released in the form of heat by controlled thermolysis, photolysis, or catalysis. However, the parent norbornadiene absorbs UV light, and the photoisomerization quantum yield is very low. Therefore, norbornadiene derivatives whose absorption is red-shifted towards the visible region of the solar spectrum are interesting research targets.
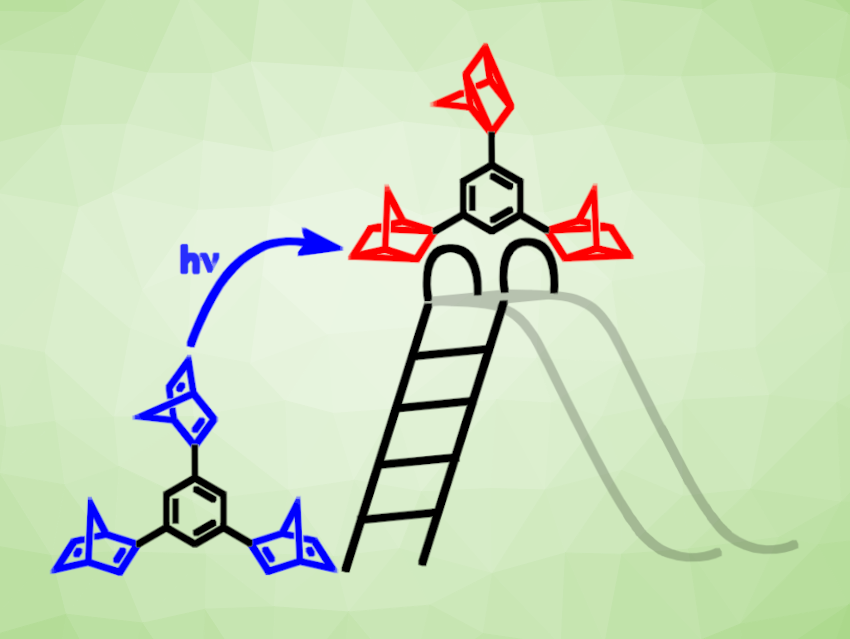
Heiko Ihmels, University of Siegen, Germany, and colleagues have developed phenyl- and naphthyl-linked bis- and tris-norbornadienes (example pictured) that can serve as promising MOST systems with very high energy densities. The team synthesized the desired compounds using Suzuki-Miyaura coupling reactions and investigated their absorption properties.
The norbornadiene derivatives showed absorption onsets of up to 386 nm and photoisomerization quantum yields of up to 56 %. The resulting quadricyclane products have half-lives of up to 14 d and very high energy densities of up to 734 KJ/Kg for a tris-quadriyclane (pictured in red). According to the researchers, these values are the best ones obtained for aryl-substituted norbornadienes. Overall, the products fulfill the necessary criteria for a MOST system and can, therefore, provide a basis for the development of materials for efficient solar energy conversion and storage.
- Bis‐ and Tris‐norbornadienes with High Energy Densities for Efficient Molecular Solar Thermal Energy Storage,
Robin Schulte, Sandra Afflerbach, Thomas Paululat, Heiko Ihmels,
Angew. Chem. Int. Ed. 2023.
https://doi.org/10.1002/anie.202309544