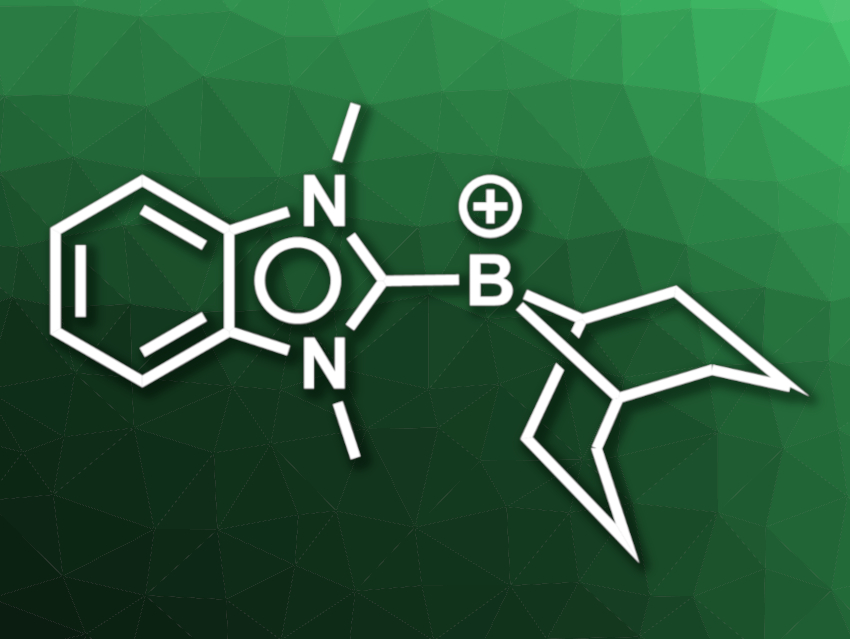
Borenium ions, i.e., molecules with a positively charged three-coordinate boron center, are interesting research targets, e.g., for metal-free catalysis. There are limited examples of borenium ions that can be used for catalytic hydrogenation reactions, mostly under very high H2 pressures. Such borenium ions are usually derived from N-heterocyclic carbenes (NHCs) such as imidazolylidenes. Replacing the imidazolylidene groups with benzimidazolylidenes could give more reactive borenium ions due to the weaker donor properties of this type of NHC. However, benzimidazolylidene-stabilized borenium ions had not been reported so far.
Swapan K Pati, Jawaharlal Nehru Centre For Advanced Scientific Research, Bangalore, India, Swadhin K. Mandal, Indian Institute of Science Education and Research (IISER) Kolkata, Mohanpur, India, and colleagues have synthesized the first benzimidazolylidene-stabilized borenium ion (pictured). The team started from 1,3-dimethyl-1H-benzo[d]imidazol-3-ium iodide, which was reacted with 9-borabicyclo[3.3.1]nonane (9-BBN) in the presence of potassium bis(trimethylsilyl)amide (KHMDS) in tetrahydrofuran (THF). The resulting benzimidazolylidene–9-BBN adduct was then reacted with trityl tetrakis(pentafluorophenyl)borate [Ph3C][B(C6F5)4] to obtain the desired benzimidazolylidene-stabilized borenium ion in the form of its [B(C6F5)4]− salt.
The researchers investigated the catalytic activity of the prepared borenium ion, using the hydrogenation of 2-phenyl quinoline as a model reaction. The boreniumWith a 10 mol% loading of the borenium ion and 1 atm of H2, they obtained the hydrogenated product 2-phenyl-1,2,3,4-tetrahydroquinoline in an isolated yield of 74 % for a reaction performed at room temperature. This reaction was extended to a variety of substituted quinolines, as well as to the synthesis of pharmaceutically active molecules.
- Benzimidazolylidene-Stabilized Borenium Ion for Catalytic Hydrogenation of N-Heterocycles,
Subir Maji, Pallavi Sarkar, Arpan Das, Swapan K. Pati, Swadhin K. Mandal,
Inorg. Chem. 2022.
https://doi.org/10.1021/acs.inorgchem.2c01841