Molecular fluorophores can have applications, e.g., in laser dyes or bioimaging probes and as electroluminescent compounds in optoelectronics. They can have excellent optical properties, such as high photoluminescence quantum yields and tunable emission wavelengths. Imidazopyridine units can be useful in small-molecule emitters. While imidazo[1,5-a]pyridines are well-investigated as imidazopyridine isomers with optical properties, studies of imidazo[1,2-a]pyridine derivatives have been limited. Emitters with an imidazo[1,2-a]pyridine core have been found to be suitable as deep-blue fluorophores. This might be useful, e.g., in organic light-emitting diodes (OLEDs).
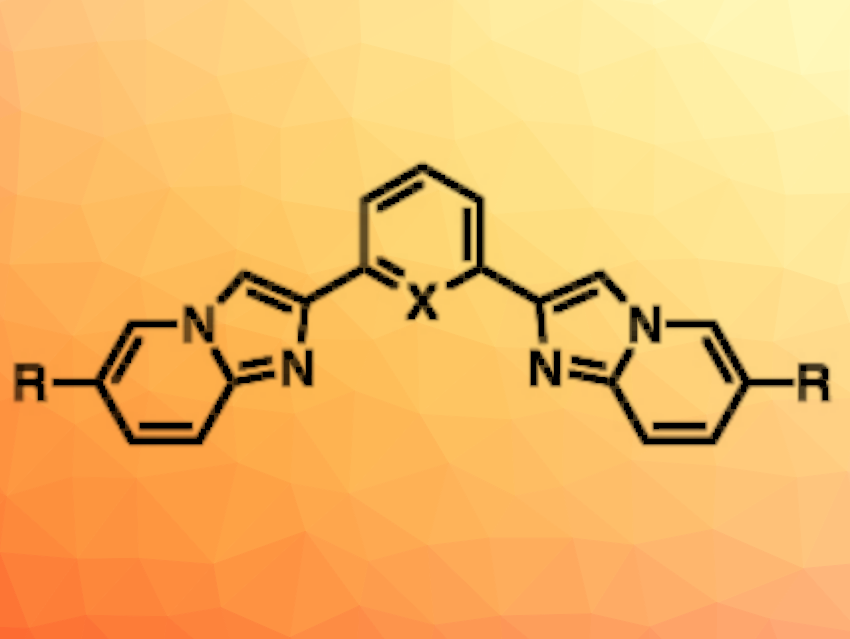
Matteo Mauro, Université de Strasbourg, CNRS, France, and colleagues have developed and synthesized a new family of bright small-molecule fluorophores with two imidazo[1,2-a]pyridine units each (general structure pictured), connected by a phenyl (X = CH) or a pyridine (X = N) bridge. The imidazopyridine units are functionalized with either electron-accepting or electron-donating peripheral groups (R = e.g., Me, C6H4(p-OMe), C6H4NPh2). This functionalization allowed the team to tune the photoluminescence.
The desired fluorophores were synthesized starting from α,α′-dibromo-1,3-diacetylbenzene or α,α′-dibromo-2,6-diacetylpyridine, which were condensed with 2-amino-5-methylpyridine or 2-amino-5-bromopyridine. The bromine atom also allowed further functionalization with peripheral substituents R, leading to two series of either phenyl or pyridine-bridged fluorophores. The researchers found that the emitters display intense fluorescence in the near-UV to deep-blue regions. This could make them useful for deep-blue electroluminescent devices, e.g., in OLEDs. They might also have applications as optical probes and molecular sensors.
- Bright V‐Shaped bis‐Imidazo[1,2‐a]pyridine Fluorophores with Near‐UV to Deep‐Blue Emission,
Elise Jouaiti, Valerio Giuso, Damien Cianfarani, Nathalie Kyritsakas, Christophe Gourlaouen, Matteo Mauro,
Chem. Asian J. 2022.
https://doi.org/10.1002/asia.202200903