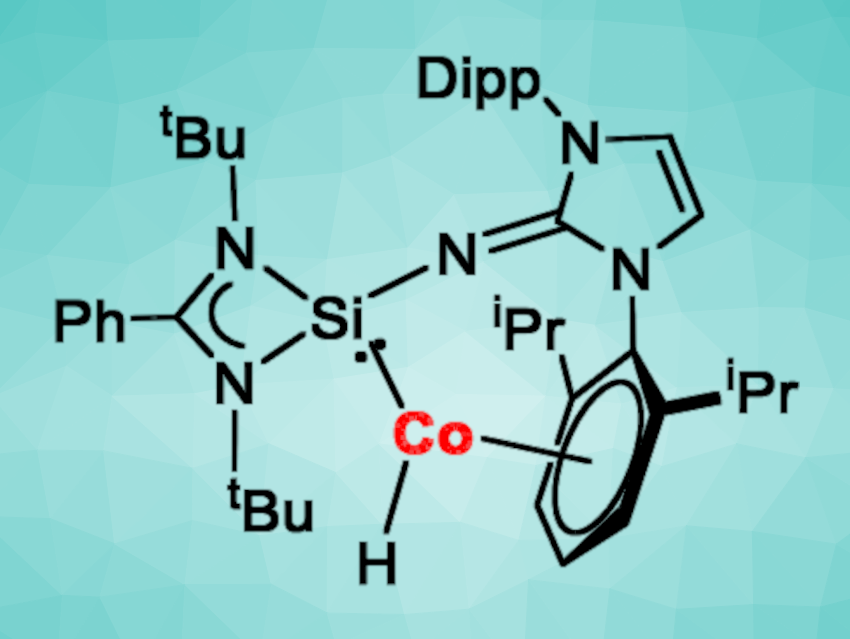
The hydrogenation of olefins to give the corresponding alkanes is an important type of reaction. The development of environmentally benign catalysts for this transformation using earth-abundant metals like cobalt is an interesting research target.
Cobalt carbonyl complexes can promote the hydrogenation of olefins, but these catalysts generally suffer from low catalytic activity and a limited substrate scope. Ligand design could help to improve these properties. For example, N-heterocyclic silylenes (NHSi), which can be considered homologues of the widely used N-heterocyclic carbenes, can be effective ligands in homogeneous catalysis.
Chuangchuang Xu, Zhenbo Mo, Nankai University, Tianjin, China, and colleagues have synthesized a cobalt(I) hydride complex with an N-heterocyclic, imino-substituted silylene (pictured) for the hydrogenation of olefins. The silylene was prepared via a reaction of LSiCl (L = PhC(NtBu)2) with NHILi (NHI = bis(2,6-(diisopropylphenyl)imidazolin-2-imino group) in toluene. The ligand was then reacted with CoCl2 and Na/Hg in tetrahydrofuran (THF) at room temperature to obtain the complex [Co(L(NHI)Si)Cl]. The desired cobalt(I) hydride complex [Co(L(NHI)Si)H] was then prepared via a reaction with KsBu3BH.
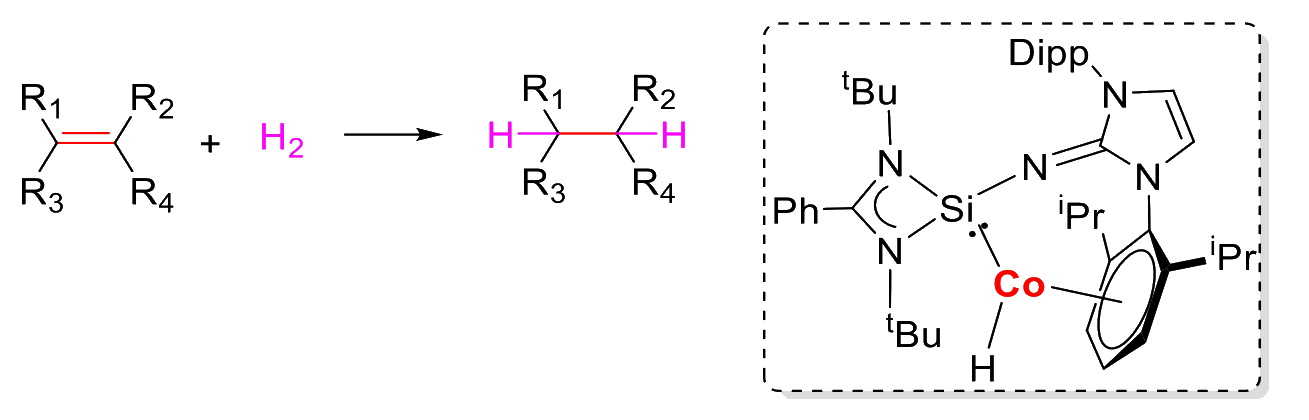
The complex exhibits high catalytic efficiency in the hydrogenation of olefins (pictured above) with good chemoselectivity and functional group compatibility. Olefins with carbonyl groups could be selectively converted to alkyl esters and ketones. According to the researchers, this type of NHSi ligand could be useful for the design of new catalysts.
- Hydrogenation of Olefins Catalyzed by a Cobalt(I) Hydride Complex with N‐Heterocyclic Silylene,
Hongwei Jia, Shaozhi Du, Chuangchuang Xu, Zhenbo Mo,
Eur. J. Inorg. Chem. 2023.
https://doi.org/10.1002/ejic.202300086