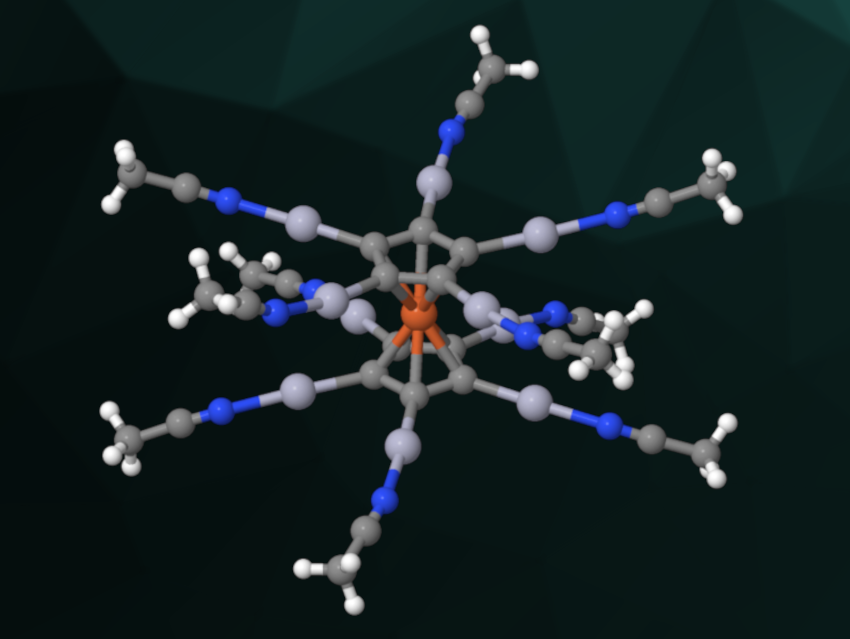
Dendrimers with redox-active building blocks can be useful, e.g., as multielectron redox catalysts or for use in electrochemical sensors. Their molecular structure is often based on a star-like shape, with a core surrounded by ligands. One widely used redox-active building block is the ferrocenyl group, but examples of “metallostars ” with a ferrocene unit in the center are rare. This might, in part, be due to the challenges involved in the perfunctionalization of ferrocenes.
Moritz Malischewski, Free University of Berlin, Germany, and colleagues have prepared decacationic metallostars with a ferrocene core via a reaction of the permercurated ferrocene FeC10(HgO2CCF3)10 with superacidic (C5F5NH)(SbF6), i.e., protonated pentafluoropyridine. They obtained [FeC10Hg10(NC5F5)n][SbF6]10. In this product, the pentafluoropyridine ligands can be easily displaced by acetonitrile (MeCN, cation pictured) or tetrahydrothiophene (THT). This improved the solubility of the compounds and allowed the researchers to fully characterize the products.
The team performed cyclic voltammetry measurements of [FeC10(Hg(MeCN))10]10+ and [FeC10(Hg(THT))10]10+ to understand the electrochemical properties of the metallostars. They found evidence for a reversible one-electron oxidation of Fe(II) to Fe(III). According to the researchers, the developed electrophilic metallostars are promising building blocks for the synthesis of larger star-shaped molecules (e.g., dendritic structures) with a robust, tenfold functionalized ferrocene core.
- A Decacationic Ferrocene-Based Metallostar,
Susanne M. Rupf, Amina Leoni Moshtaha, Moritz Malischewski,
Chem. Sci. 2023.
https://doi.org/10.1039/D2SC06151A