Click chemistry has become an important tool for chemists, as it can be used to prepare compounds quickly in an efficient manner by joining small units together. This method is particularly helpful in the preparation of triazoles, natural products, pharmaceuticals and modified peptides.
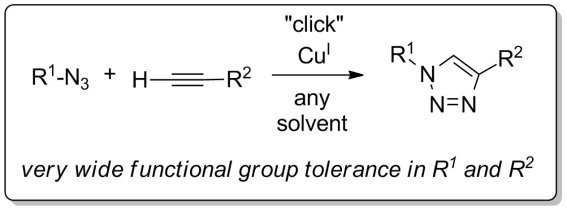
One of the most popular reactions within the click chemistry realm is the copper-catalyzed alkyne–azide cycloaddition reaction. Its importance in today’s preparative methods is exemplified by the microreview CuI-Catalyzed Alkyne-Azide “Click” Cycloadditions from a Mechanistic and Synthetic Perspective by Victoria Bock, Henk Hiemstra, and Jan van Maarseveen, University of Amsterdam, the Netherlands.
This article has now received 503 citations (ISI Web of Science, 18.02.2011). This is by far the most-cited article of all articles published in 2006 in the subject category “Chemistry, Organic”, according to ISI Web of Science.
Henk Hiemstra serves on the Editorial Board of European Journal of Organic Chemistry (EurJOC) and has been instrumental in its development over the years.
The EurJOC team congratulates Hiemstra and his co-workers for this great success.
Images: (c) Wiley-VCH
- CuI-Catalyzed Alkyne–Azide “Click” Cycloadditions from a Mechanistic and Synthetic Perspective
V. D. Bock, H. Hiemstra, J. H. van Maarseveen,
Eur. J. Org. Chem. 2006, 51–68.
DOI: 10.1002/ejoc.200500483