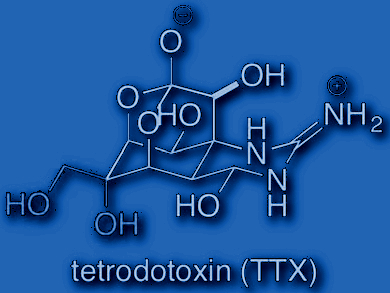
Tohru Fukuyama, Nagoya University, Japan, and colleagues have developed a synthetic pathway to (–)-tetrodotoxin (TTX), a neurotoxin from Japanese fugu, and its natural congener, 11-norTTX-6(R)-ol. TTX works as a potent inhibitor of voltage-gated sodium ion channels (so-called Navs).
Since Nav channels are responsible for initiation and propagation of action potentials, their disorders cause a wide range of serious diseases like epilepsy and neuropathic pain. Despite a long history of Nav investigation, the molecular mechanism of its behavior remains a mystery. Because of its selectivity and potency against Navs, TTX and its analogues could serve as invaluable tools for Nav research.
The team established an enantioselective route to TTX and also applied it to reach the scarcely available TTX congener 11-norTTX-6(R)-ol via a common intermediate. The synthesis starts from the commercially available p-benzoquinone and includes a [3,3]-sigmatropic rearrangement of an allylic cyanate, and intramolecular 1,3-dipolar cycloaddition of a nitrile oxide. Neutral deprotection at the final step enabled easy purification of the products. Overall, the synthesis has 31 steps and, according to the researchers, should be applicable to the synthesis of a variety of natural and unnatural TTX congeners.
- Total Synthesis of (–)-Tetrodotoxin and 11-norTTX-6(R)-ol,
Tomoaki Maehara, Keisuke Motoyama, Tatsuya Toma, Satoshi Yokoshima, Tohru Fukuyama,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201611574