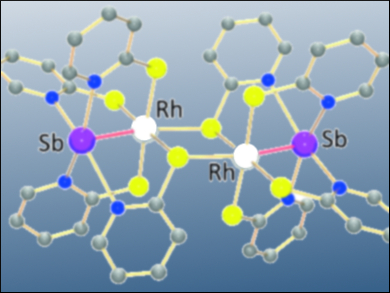
Heterobinuclear complexes are interesting because the nature of the bond between the two metal centers can determine the complex’s potential for catalysis or sensing applications. Several types of such complexes have already been characterized, including some that contain transition metal (TM) and main group metals (E). These bonds could either be covalent (TM–E) or dative of either Z- (TM→E) or L-types (TM←E). The bridging ligands and oxidation states of the metal centers may also affect the nature of these bonds.
Manuel Iglesias, University of Zaragoza, Spain, Jörg Wagler, Technische Universität Bergakademie Freiberg, Germany, and colleagues synthesized paddlewheel-shaped heterobinuclear complexes with rhodium and antimony metal centers and pyridine-2-thiolate (pyS) ligands (pictured above). They also oxidized the complex and characterized both forms using single-crystal X-ray diffraction and 121Sb Mössbauer spectroscopy. The team also used density functional theory (DFT) calculations to gain insight on the nature of the Rh–Sb bond.
The team showed that although the 121Sb Mössbauer spectroscopy indicates similar contributions of both metals to the bond, it may not be entirely covalent in nature. DFT calculations showed that there are some electronic features that show Rh→Sb (Z-type) and Rh←Sb (L-type) characteristics for the reduced and oxidized forms, respectively. The bonding situation can thus be explained by different resonance structures (pictured below, oxidized form on the right). According to the team, these flexible characteristics of similar bonds makes heterobinuclear complexes interesting research targets.

- Synthesis and Oxidation of a Paddlewheel-Shaped Rhodium/Antimony Complex Featuring Pyridine-2-Thiolate Ligands,
Erik Wächtler, Luis A. Oro, Manuel Iglesias, Birgit Gerke, Rainer Pöttgen, Robert Gericke, Jörg Wagler,
Chem. Eur. J. 2017.
DOI: 10.1002/chem.201605485