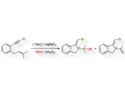
Organic luminophores are particularly attractive because of their wide application scope in various fields. A subtle modification of their molecular skeleton can pronouncedly change their luminescence.
Yulan Chen, Tianjin University, China, and colleagues successfully tuned the photophysical properties of luminophores based on 9-styrylanthracene (pictured) via geometry control at the molecular scale. These luminophores were synthesized via a Horner–Wadsworth–Emmons reaction. The cubic anchoring terminal groups on styrylanthracene effectively hinder intermolecular packing, giving highly luminescent materials in both aggregated and solid state.
The synthesized materials can be utilized to detect picric acid (a model compound for explosives) via fluorescence quenching. More importantly, these luminophores showed reversible solid-state mechanofluorochromic properties, and their mechano-induced emission shifts were highly dependent on molecular symmetry.
- Structural Insights into 9-Styrylanthracene-Based Luminophores: Geometry Control vs Mechanofluorochromism and Sensing Properties,
Xu Zhang, Yi-Xuan Wang, Jing Zhao, Pengfei Duan, Yulan Chen, Long Chen,
Chem. Asian J. 2017.
DOI: 10.1002/asia.201700183