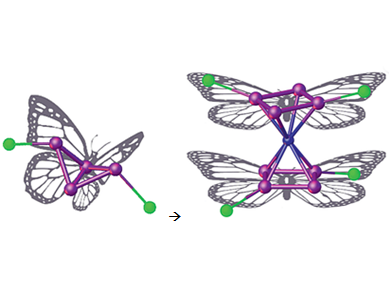
The selective activation of white phosphorus (P4) is a popular research target, as phosphorus is an important starting material in the chemical industry. One of the first degradation steps in the reaction of P4 with metal fragments results in the formation of P4 “butterfly” complexes (pictured left; [{Cp′′′Fe(CO)2}2(μ,η1:1-P4)] (Cp′′′=η5-C5H2tBu3)). These structures have been shown to act as a new all-phosphorus ligand in coordination chemistry, with properties comparable to the popular ligand 1,1-bis(diphenylphosphino)methane (dppm) [1].
Manfred Scheer, University of Regensburg, Germany, and colleagues have shown that these complexes can react with Lewis acids with either retention or isomerization of the P4 butterfly unit. The outcome can be controlled by the nature of the Lewis acid. Structural retention of the P4 unit is observed when the complex is reacted with [FeBr2∙dme] (dme = dimethoxyethane).
When the butterfly unit is reacted with iron(II) complexes containing only weakly coordinating ligands, such as [Fe(CH3CN)6][PF6]2, an isomerization of the butterfly complex takes place. The resulting homoleptic-like iron-phosphorus product contains unprecedented aromatic 6π-electron R2P4 ligands. It is the first fully characterized “carbon-free” sandwich complex of this kind (pictured right). This isomerization represents a so far unreported step in P4 activation.
- Rearrangement of a P4 Butterfly Complex—The Formation of a Homoleptic Phosphorus-Iron Sandwich Complex,
Julian Müller, Sebastian Heinl, Christoph Schwarzmaier, Gábor Balázs, Martin Keilwerth, Karsten Meyer, Manfred Scheer,
Angew. Chem. Int. Ed. 2017.
DOI: 10.1002/anie.201703175
Reference
- [1] E4 Butterfly Complexes (E = P, As) as Chelating Ligands,
Christoph Schwarzmaier, Sebastian Heinl, Gábor Balázs, Manfred Scheer,
Angew. Chem. Int. Ed. 2015, 54, 13116–13121.
DOI: 10.1002/anie.201506784