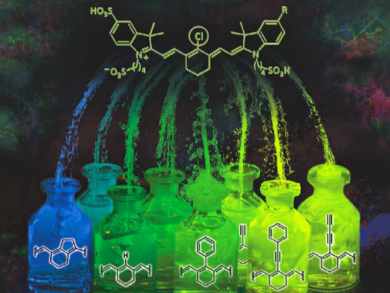
Indotricarbocyanine probes are among the fluorescent molecules that are of particular interest for bioimaging. Unfortunately, synthetic routes towards these dyes have issues with chemical stability and practicality.
Roland Krämer and Sandra G. König, University of Heidelberg, Germany, have developed a series of near-infrared (NIR) fluorescent probes which are coupled to folate. The researchers used symmetric and asymmetric backbones based on indotricarbocyanine as well as various reaction strategies (coupling reactions, dehalogenation, and substitution reactions) to functionalize the central carbon (substituted with Cl in the picture).
The team evaluated the photophysical properties as well as the stability of the dyes. Additionally, they attached a folate molecule to their NIR dyes via a COOH substituent. Many cancer cell types overexpress folate receptors. These receptors are often exploited to selectively target fluorescent probes and drug delivery systems to tumors and cancer cells. The researchers used confocal microscopy and flow cytometry to show that the folate-containing fluorescent probes stain HeLa cancer cells, but not healthy fibroblasts.
According to the team, this synthetic concept to structurally diverse probes may support the further development and the clinical adoption of this class of NIR tracers.
- Accessing Structurally Diverse Near-Infrared Cyanine Dyes for Folate Receptor-Targeted Cancer Cell Staining,
Sandra G. König, Roland Krämer,
Chem. Eur. J. 2017.
DOI: 10.1002/chem.201700026