Increasing water pollution, especially by organic compounds such as dyes, pesticides, and fertilizers, has made the removal of such pollutants a hot topic in research. Traditional adsorption and coagulation technologies can remove the pollutants from water, but do not break down the compounds themselves. An alternative is the use of photocatalysts which, with the aid of sunlight, can break down pollutants into less harmful materials.
Xianhui Bu, California State University, Long Beach, USA, Pingyun Feng, University of California, Riverside, USA, and colleagues have extended the search for such photocatalysts beyond “traditional” semiconductor oxides (e.g., TiO2, ZnO, Fe2O3) to chalcogenide frameworks. These crystalline porous materials, related to zeolites, have the advantage of tunable structures. This allows the size and charge of the pore channels to be tailored towards certain target molecules.
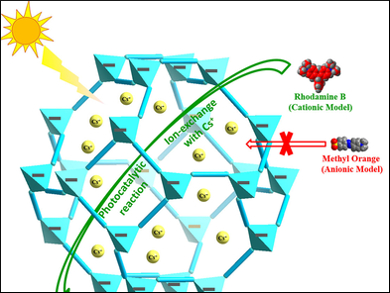
The material the team tested contains clusters containing zinc, gallium, germanium, and sulfur ([Zn0.76Ga0.70Ge2.54S8]2.22−). Dye molecules are used for testing the photocatalytic properties because a simple color change can quickly determine whether the catalyst is working. The chalcogenide framework the researchers used has positively charged Cs+ in its channels, so selectively prefers positively charged dyes such as methylene blue. Size selectivity is also possible, with smaller dyes preferred over bulky ones in this case. The work demonstrates not only that chalcogenide frameworks can be used as photocatalysts, but also that the tunability of framework structures can allow specific pollutants to be targeted.
- Selective Ion Exchange and Photocatalysis by Zeolite-Like Semiconducting Chalcogenide,
Xitong Chen, Xianhui Bu, Qipu Lin, Chengyu Mao, Quan-Guo Zhai, Yuan Wang, Pingyun Feng,
Chem. Eur. J. 2017.
DOI: 10.1002/chem.201702418