Supercapacitors are energy storage devices that bridge the gap between rechargeable batteries and electrolytic capacitors. They are useful for applications which require rapid charge/discharge cycles rather than long term energy storage. One automotive use of supercapacitors is in regenerative braking. Energy is generated by the brakes, which can be stored in the short term, and then used to provide bursts of power.
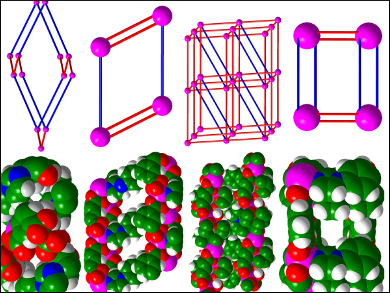
Zuo-Xi Li, Northwest University, Xi’an, China, and colleagues have prepared metal-organic frameworks (MOFs) as precursors to porous carbons that are used for supercapacitors. Initially, the team synthesized a series of different cadmium MOFs using the same starting materials, but altering the solvent. The MOFs were converted to porous carbon by heating them to 1000 °C under a nitrogen atmosphere. The different crystal structures of the Cd-MOFs (examples pictured) lead to differing levels of porosity in the calcined carbon products.
The team found that the material synthesized using dimethylformamide (DMF) exhibits the best properties with large specific capacitance (156 F g–1 at 0.5 A g–1). The DMF-derived electrode shows excellent cycling stability (ca. 84.2 % after 5000 cycles at 1 A g–1), which means it could be used as a suitable electrode material for supercapacitors.
- Solvent-Induced Cd-MOFs with Adjustable Guest-Evacuated Porosity: Application in the Controllable Assembly of MOF-Derived Porous Carbon Materials for Supercapacitors,
Zuo Xi Li, Man-Li Yue, Yi-Fan Jiang, Lin Zhang, Cheng-Yan Yu, Kang-Yu Zou,
Chem. Eur. J. 2017.
DOI: 10.1002/chem.201702694