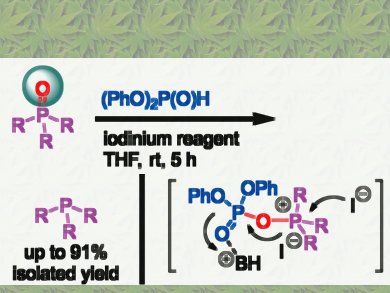
Peng Li, Raphael Wischert, and Pascal Métivier, Solvay Asia Pacific, Shanghai, China, have achieved an easy reduction of phosphine oxides to phosphines with inexpensive trialkyl phosphite at room temperature. The iodine-catalyzed reduction of various phosphine oxides to phosphines takes place in the absence of other reductants such as silanes. The mild reaction conditions, scalability, and simple purification requirements make this process a method of choice for the large-scale production and regeneration of a variety of phosphines.
Based on experiments and DFT calculations, the researchers propose a mechanism involving oxygen transfer between the starting phosphine oxide and the phosphite reagent via an iodophosphate intermediate. This highly reactive species easily undergo nucleophilic attack by the oxygen atom of triphenylphosphine oxide. This oxygen transfer reaction is remarkable in that the phosphorus atom in phosphite is considered to be more electron-deficient than that of triphenylphosphine. Thus using phosphite as a reducing reagent for phosphine oxide is not obvious.
The researchers are further investigating the reaction to better understand its scope, to optimize the reaction conditions, and fully characterize the mechanism.
- Mild Reduction of Phosphine Oxides with Phosphites To Access Phosphines,
Peng Li, Raphael Wischert, Pascal Métivier,
Angew. Chem. Int. Ed. 2017.
https://doi.org/10.1002/anie.201709519