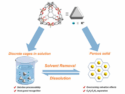
Nitrous acid (HNO2) can be used in nitrosation and nitration reactions. However, the strongly acidic and oxidizing character of nitrous acid means harsh reaction conditions, which can limit the substrate scope and cause the formation of by-products. Dinitrogen trioxide, N2O3, dissociates into nitric oxide (NO) and nitrogen dioxide (NO2) radicals (pictured) and could theoretically be used to replace nitrous acid as a reactant. Unfortunately, N2O3 is quite unstable.
Kristopher A. Rosadiuk and D. Scott Bohle, McGill University, Montreal, Canada, have used organic solvents to stabilize N2O3 and enable its use in nitrosation and nitration reactions.The team prepared N2O3 solutions by alternately passing NO gas and oxygen through a dry solvent cooled to 0 °C. The formed reagent can then directly be used in a one-pot reaction approach to prepare, e.g., benzenediazonium nitrite ([PhN2][NO2]), nitrosyl chloride (NOCl), or nitrosylsulfuric acid, (ONOSO3H).
Most aprotic solvents that are either polar or aromatic can stabilize N2O3. The team used solutions in, e.g., acetic anhydride, acetone, acetonitrile, benzene, chlorobenzene, chloroform, dichloromethane, dimethyl sulfoxide (DMSO), ethyl acetate, tetrahydrofuran (THF), and toluene. According to the researchers, N2O3 could become a useful reagent in synthesis using this solvent-stabilization approach.
- Anhydrous Dinitrogen Trioxide Solutions for Brønsted Acid Free Nitrous Acid Chemistry,
Scott Bohle, Kristopher A. Rosadiuk,
Eur. J. Inorg. Chem. 2017.
https://doi.org/10.1002/ejic.201701091