Nanoparticles with Defined Shapes and Atom Numbers
A new approach to making silver nanoparticles, and potentially other types of nanoparticles, with defined shapes and specific numbers of metal atoms has been developed by Di Sun, Shandong University, Jinan, and Xiamen University, both China, and colleagues.
Despite a growing interest in the anisotropic assembly of inorganic nanoparticles with atomically precise structures formed through self-assembly processes, such entities remain extremely rare in the scientific literature. The researchers hoped to remedy that situation by developing a general synthetic approach that would allow them to construct nanoparticles, in this case silver nanoparticles, with a definite shape and atom count.
Anisotropic Nanoclusters
It is well known that the structure of a nanoparticle closely correlates with its properties—both size and shape matter. Moreover, for the sake of scientific reproducibility and technological development of these materials, it is important to find ways to make them consistently with well-defined sizes and shapes in a batch. A spread of shapes and sizes does nothing for reliable results.
Isotropic spherical particles are almost commonplace in the nano lab, but there is also a need to modify such particles in order to control their surface features and to tune their properties, based on the number of atoms in the cluster and the faces they present to the outside world. Electrical, photonic, catalytic, and other properties might all better be defined for specific anisotropic nanoclusters.
Internal Templates
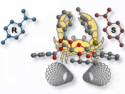
With all of this in mind, the researchers used two different molybdate anions as internal templates upon which they could build such silver nanoparticles. They used five MoO42– anions as the internal template to make a silver nanoparticle containing 52 silver atoms. They protected the surface of the resulting cluster with sulfur- and phosphorus-based ligands. In a second silver nanoparticle containing 76 silver atoms, a pair of Mo6O228– anions acted as the internal template.
The team explains how the regiospecific distribution of diphosphine ligands on the surface, connected through organic thiol bridging ligands, works synergistically to arrange multiple molybdate anions as the template during the formation of the nanocluster. This effect tailors the shape of the cluster and the number of silver atoms. The Ag52 cluster has an anisotropic oblate spheroid shape, while the Ag76 nanocluster is shaped like an elongated rod.
New Shape-Controlling Strategy
The researchers suggest that, “This work opens up new avenues for the synthesis of silver nanoclusters with novel metal skeleton shapes and anisotropic surface structures.” They add that it also provides useful insights into the anisotropic growth of such nanoclusters through their surface modification and self-assembly using templates.
“Nanoclusters and nanoparticles tend to be spherical and anisotropic shapes are difficult to coax out, yet the anisotropic ones are particularly attractive in terms of exploring new properties,” suggests Rongchao Jin, Carnegie Mellon University, Pittsburgh, PA, USA. “In this work, the team has devised a nice strategy of utilizing the synergistic effects from the molybdate-anion template and the binary ligands (i.e., thiol and diphosphine). This work not only leads to two anisotropic nanoclusters, but also offers a new strategy for controlling particle shape through interior anion organization and surface ligand modification.”
- Anisotropic Assembly of Ag52 and Ag76 Nanoclusters,
Jia-Wei Liu, Lei Feng, Hai-Feng Su, Zhi Wang, Quan-Qin Zhao, Xing-Po Wang, Chen-Ho Tung, Di Sun, Lan-Sun Zheng,
J. Am. Chem. Soc. 2018.
https://doi.org/10.1021/jacs.7b12777